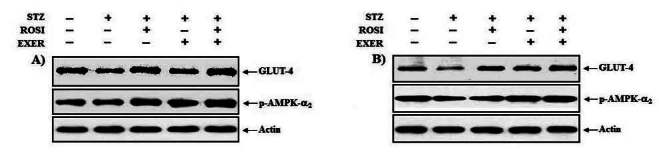
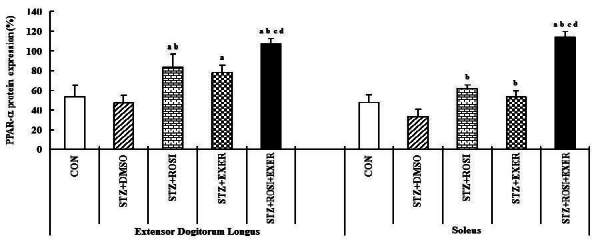
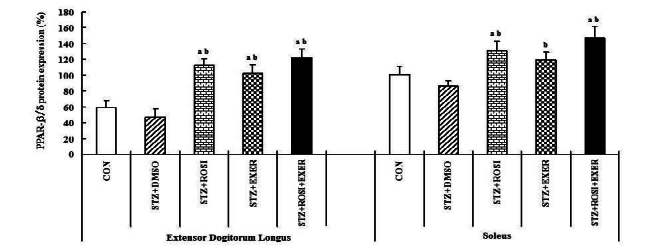
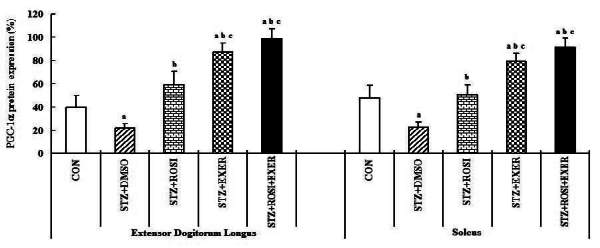
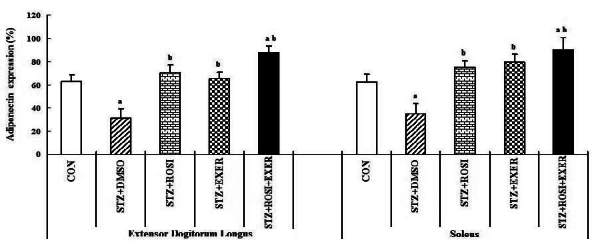
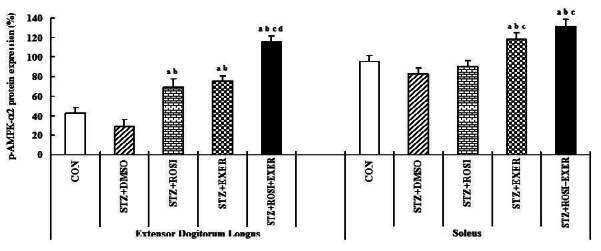
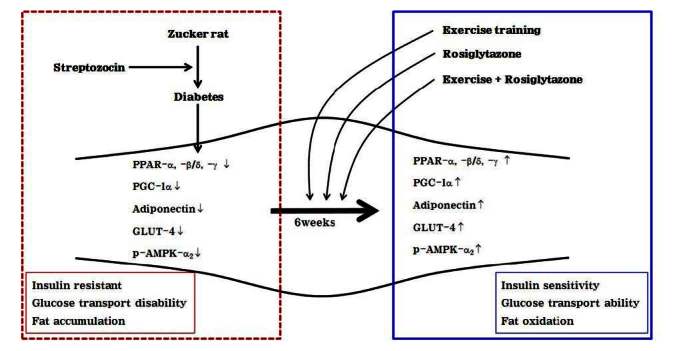
1.
Aragno M, Mastrocola R, Catalano MG, Brignardello E, Danni O, Boccuzzi. Oxidative stress impairs skeletal muscle repair in diabetic rats.
Diabetes G. 2004;53:1082-8.

Aragno M., Mastrocola R., Catalano MG., Brignardello E., Danni O., Boccuzzi. . Oxidative stress impairs skeletal muscle repair in diabetic rats.
Diabetes G 2004;53:1082-8. PMID:
10.2337/diabetes.53.4.1082.
2.
Arany Z. PGC-1 coactivators and skeletal muscle adaptations in health and disease.
Curr Opin Genet Dev. 2008;18:426-34.

Arany Z.. PGC-1 coactivators and skeletal muscle adaptations in health and disease.
Curr Opin Genet Dev 2008;18:426-34. PMID:
10.1016/j.gde.2008.07.018. PMID:
18782618.
3.
Bell GI, Kayano T, Buse JB, Burant CF, Takeda J, Lin D, Fukumoto H, Seino S. Molecular biology of mammalian glucose transporters.
Diabetes Care. 1990;13:198-208.

Bell GI., Kayano T., Buse JB., Burant CF., Takeda J., Lin D., Fukumoto H., Seino S.. Molecular biology of mammalian glucose transporters.
Diabetes Care 1990;13:198-208. PMID:
10.2337/diacare.13.3.198. PMID:
2407475.
4.
Blüher M, Brennan AM, Kelesidis T, Kratzsch J, Fasshauer M, Kralisch S, Williams CJ, Mantzoros CS. Total and high-molecular weight adiponectin in relation to metabolic variables at baseline and in response to an exercise treatment program: comparative evaluation of three assays.
Diabetes Care. 2007; 30:280-5.

Blüher M., Brennan AM., Kelesidis T., Kratzsch J., Fasshauer M., Kralisch S., Williams CJ., Mantzoros CS.. Total and high-molecular weight adiponectin in relation to metabolic variables at baseline and in response to an exercise treatment program: comparative evaluation of three assays.
Diabetes Care 2007;30:280-5. PMID:
10.2337/dc06-1362. PMID:
17259495.
5.
Blüher M, Bullen JW Jr, Lee JH, Kralisch S, Fasshauer M, Klöting N, Niebauer J, Schön MR, Williams CJ, Mantzoros CS. Circulating adiponectin and expression of adiponectin receptors in human skeletal muscle: associations with metabolic parameters and insulin resistance and regulation by physical training.
J Clin Endocrinol Metab. 2006;91:2310-6.

Blüher M., Bullen JW. Jr, Lee JH., Kralisch S., Fasshauer M., Klöting N., Niebauer J., Schön MR., Williams CJ., Mantzoros CS.. Circulating adiponectin and expression of adiponectin receptors in human skeletal muscle: associations with metabolic parameters and insulin resistance and regulation by physical training.
J Clin Endocrinol Metab 2006;91:2310-6. PMID:
10.1210/jc.2005-2556. PMID:
16551730.
6.
Cha BS, Ciaraldi TP, Park KS, Carter L, Mudaliar SR, Henry RR. Impaired fatty acid metabolism in type 2 diabetic skeletal muscle cells is reversed by PPARgamma agonists. Am J Physiol Endocrinol Meta. 2005;289:E151-9.

Cha BS., Ciaraldi TP., Park KS., Carter L., Mudaliar SR., Henry RR.. Impaired fatty acid metabolism in type 2 diabetic skeletal muscle cells is reversed by PPARgamma agonists.
Am J Physiol Endocrinol Meta 2005;289:E151-9. PMID:
10.1152/ajpendo.00141.2004.
7.
Chen MB, McAinch AJ, Macaulay SL, Castelli LA, O’brien PE, Dixon JB, Cameron-Smith D, Kemp BE, Steinberg G. Impaired activation of AMP-kinase and fatty acid oxidation by globular adiponectin in cultured human skeletal muscle of obese type 2 diabetics.
J Clin Endocrinol Meta. 2005;90:3665-72.

Chen MB., McAinch AJ., Macaulay SL., Castelli LA., O’brien PE., Dixon JB., Cameron-Smith D., Kemp BE., Steinberg G.. Impaired activation of AMP-kinase and fatty acid oxidation by globular adiponectin in cultured human skeletal muscle of obese type 2 diabetics.
J Clin Endocrinol Meta 2005;90:3665-72. PMID:
10.1210/jc.2004-1980.
8.
DeFronzo RA, Bonadonna RC, Ferrannini E. Pathogenesis of NIDDM. A balanced overview.
Diabetes Care. 1992;15:318-68.

DeFronzo RA., Bonadonna RC., Ferrannini E.. Pathogenesis of NIDDM. A balanced overview.
Diabetes Care 1992;15:318-68. PMID:
10.2337/diacare.15.3.318. PMID:
1532777.
9.
Finck BN, Kelly DP. PGC-1 coactivators: inducible regulators of energy metabolism in health and disease.
J Clin Invest. 2006;116:615-22.

Finck BN., Kelly DP.. PGC-1 coactivators: inducible regulators of energy metabolism in health and disease.
J Clin Invest 2006;116:615-22. PMID:
10.1172/JCI27794. PMID:
16511594.
10.
Fonseca V. Effect of thiazolidinediones on body weight in patients with diabetes mellitus. Am J Med. 2003;115:42-8.

Fonseca V.. Effect of thiazolidinediones on body weight in patients with diabetes mellitus.
Am J Med 2003;115:42-8. PMID:
10.1016/j.amjmed.2003.09.005.
11.
Gilde AJ, Van Bilsen M. Peroxisome proliferator-activated receptors (PPARS): regulators of gene expression in heart and skeletal muscle.
Acta Physiol Scand. 2003;178:425-34.

Gilde AJ., Van Bilsen M.. Peroxisome proliferator-activated receptors (PPARS): regulators of gene expression in heart and skeletal muscle.
Acta Physiol Scand 2003;178:425-34. PMID:
10.1046/j.1365-201X.2003.01161.x. PMID:
12864748.
12.
Hardie DG, Hawley SA. AMP-activated protein kinase: the energy charge hypothesis revisited.
Bioessays. 2001;23:1112-9.

Hardie DG., Hawley SA.. AMP-activated protein kinase: the energy charge hypothesis revisited.
Bioessays 2001;23:1112-9. PMID:
10.1002/bies.10009. PMID:
11746230.
13.
Hevener AL, Reichart D, Olefsky J. Exercise and thiazolidinedione therapy normalize insulin action in the obese Zucker fatty rat.
Diabetes. 2000;49:2154-9.

Hevener AL., Reichart D., Olefsky J.. Exercise and thiazolidinedione therapy normalize insulin action in the obese Zucker fatty rat.
Diabetes 2000;49:2154-9. PMID:
10.2337/diabetes.49.12.2154. PMID:
11118020.
14.
Hotta K, Funahashi T, Bodkin NL, Ortmeyer HK, Arita Y, Hansen BC, Matsuzawa Y. Circulating concentrations of the adipocyte protein adiponectin are decreased in parallel with reduced insulin sensitivity during the progression to type 2 diabetes in rhesus monkeys.
Diabetes. 2001;50:1126-33.

Hotta K., Funahashi T., Bodkin NL., Ortmeyer HK., Arita Y., Hansen BC., Matsuzawa Y.. Circulating concentrations of the adipocyte protein adiponectin are decreased in parallel with reduced insulin sensitivity during the progression to type 2 diabetes in rhesus monkeys.
Diabetes 2001;50:1126-33. PMID:
10.2337/diabetes.50.5.1126. PMID:
11334417.
15.
Jucker BM, Schaeffer TR, Haimbach RE, Mayer ME, Ohlstein DH, Smith SA, Cobitz AR, Sarkar SK. Reduction of intramyocellular lipid following short-term rosiglitazone treatment in Zucker fatty rats: an in vivo nuclear magnetic resonance study.
Metabolism. 2003;52:218-25.

Jucker BM., Schaeffer TR., Haimbach RE., Mayer ME., Ohlstein DH., Smith SA., Cobitz AR., Sarkar SK.. Reduction of intramyocellular lipid following short-term rosiglitazone treatment in Zucker fatty rats: an in vivo nuclear magnetic resonance study.
Metabolism 2003;52:218-25. PMID:
10.1053/meta.2003.50040. PMID:
12601636.
16.
Kelley DE, He J, Menshikova EV, Ritov VB. Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes.
Diabetes. 2002;51:2944-50.

Kelley DE., He J., Menshikova EV., Ritov VB.. Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes.
Diabetes 2002;51:2944-50. PMID:
10.2337/diabetes.51.10.2944. PMID:
12351431.
17.
Koves TR, Li P, An J, Akimoto T, Slentz D, Ilkayeva O, Dohm GL, Yan Z, Newgard CB, Muoio DM. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency.
J Biol Chem. 2005;280:33588-98.

Koves TR., Li P., An J., Akimoto T., Slentz D., Ilkayeva O., Dohm GL., Yan Z., Newgard CB., Muoio DM.. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency.
J Biol Chem 2005;280:33588-98. PMID:
10.1074/jbc.M507621200. PMID:
16079133.
18.
Kramer D, Shapiro R, Adler A, Bush E, Rondinone CM. Insulin-sensitizing effect of rosiglitazone (BRL-49653) by regulation of glucose transporters in muscle and fat of Zucker rats.
Metabolism. 2001;50:1294-300.

Kramer D., Shapiro R., Adler A., Bush E., Rondinone CM.. Insulin-sensitizing effect of rosiglitazone (BRL-49653) by regulation of glucose transporters in muscle and fat of Zucker rats.
Metabolism 2001;50:1294-300. PMID:
10.1053/meta.2001.27202. PMID:
11699047.
19.
LeBrasseur NK, Ruderman NB. Why might thiazolidinediones increase exercise capacity in patients with type 2 diabetes?
Diabetes Care. 2005;28:2975-7.

LeBrasseur NK., Ruderman NB.. Why might thiazolidinediones increase exercise capacity in patients with type 2 diabetes?
Diabetes Care 2005;28:2975-7. PMID:
10.2337/diacare.28.12.2975. PMID:
16306565.
20.
Lessard SJ, Rivas DA, Chen ZP, Bonen A, Febbraio MA, Reeder DW, Kemp BE, Yaspelkis BB , Hawley JA. Tissue-specific effects of rosiglitazone and exercise in the treatment of lipid-induced insulin resistance.
Diabetes. 2007;56:1856-64.

Lessard SJ., Rivas DA., Chen ZP., Bonen A., Febbraio MA., Reeder DW., Kemp BE., Yaspelkis BB., Hawley JA.. Tissue-specific effects of rosiglitazone and exercise in the treatment of lipid-induced insulin resistance.
Diabetes 2007;56:1856-64. PMID:
10.2337/db06-1065. PMID:
17440174.
21.
Lessard SJ, Lo Giudice SL, Lau W, Reid JJ, Turner N, Febbraio MA, Hawley JA, Watt MJ. Rosiglitazone enhances glucose tolerance by mechanisms other than reduction of fatty acid accumulation within skeletal muscle.
Endocrinology. 2004;145:5665-70.


Lessard SJ., Lo Giudice SL., Lau W., Reid JJ., Turner N., Febbraio MA., Hawley JA., Watt MJ.. Rosiglitazone enhances glucose tolerance by mechanisms other than reduction of fatty acid accumulation within skeletal muscle.
Endocrinology 2004;145:5665-70. PMID:
10.1210/en.2004-0659. PMID:
15375026.
22.
Lihn AS, Pedersen SB, Richelsen B. Adiponectin: action, regulation and association to insulin sensitivity.
Obes Rev. 2005;6: 13-21.

Lihn AS., Pedersen SB., Richelsen B.. Adiponectin: action, regulation and association to insulin sensitivity.
Obes Rev 2005;6:13-21. PMID:
10.1111/j.1467-789X.2005.00159.x. PMID:
15655035.
23.
Luciano E, Carneiro EM, Carvalho CR, Carvalheira JB, Peres SB, Reis MA, Saad MJ, Boschero AC, Velloso LA. Endurance training improves responsiveness to insulin and modulates insulin signal transduction through the phosphatidylinositol 3-kinase/Akt-1 pathway.
Eur J Endocrinol. 2002;147:149-57.

Luciano E., Carneiro EM., Carvalho CR., Carvalheira JB., Peres SB., Reis MA., Saad MJ., Boschero AC., Velloso LA.. Endurance training improves responsiveness to insulin and modulates insulin signal transduction through the phosphatidylinositol 3-kinase/Akt-1 pathway.
Eur J Endocrinol 2002;147:149-57. PMID:
10.1530/eje.0.1470149. PMID:
12088932.
24.
Mastrocola R, Reffo P, Penna F, Tomasinelli CE, Boccuzzi G, Baccino FM, Aragno M, Costelli P. Muscle wasting in diabetic and in tumor-bearing rats: role of oxidative stress.
Free Radic Biol Med. 2008;44:584-93.

Mastrocola R., Reffo P., Penna F., Tomasinelli CE., Boccuzzi G., Baccino FM., Aragno M., Costelli P.. Muscle wasting in diabetic and in tumor-bearing rats: role of oxidative stress.
Free Radic Biol Med 2008;44:584-93. PMID:
10.1016/j.freeradbiomed.2007.10.047. PMID:
18053817.
25.
Mensink M, Hesselink MK, Russell AP, Schaart G, Sels JP, Schrauwen P. Improved skeletal muscle oxidative enzyme activity and restoration of PGC-1 alpha and PPAR beta/delta gene expression upon rosiglitazone treatment in obese patients with type 2 diabetes mellitus.
Int J Obes (Lond). 2007; 31:1302-10.


Mensink M., Hesselink MK., Russell AP., Schaart G., Sels JP., Schrauwen P.. Improved skeletal muscle oxidative enzyme activity and restoration of PGC-1 alpha and PPAR beta/delta gene expression upon rosiglitazone treatment in obese patients with type 2 diabetes mellitus.
Int J Obes (Lond) 2007;31:1302-10. PMID:
10.1038/sj.ijo.0803567. PMID:
17310221.
26.
Mootha VK, Lindgren CM, Eriksson KF, Subramanian A, Sihag S, Lehar J, Puigserver P, Carlsson E, Ridderstråle M, Laurila E, Houstis N, Daly MJ, Patterson N, Mesirov JP, Golub TR, Tamayo P, Spiegelman B, Lander ES, Hirschhorn JN, Altshuler D, Groop LC. PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes.
Nat Genet. 2003;34:267-73.


Mootha VK., Lindgren CM., Eriksson KF., Subramanian A., Sihag S., Lehar J., Puigserver P., Carlsson E., Ridderstråle M., Laurila E., Houstis N., Daly MJ., Patterson N., Mesirov JP., Golub TR., Tamayo P., Spiegelman B., Lander ES., Hirschhorn JN., Altshuler D., Groop LC.. PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes.
Nat Genet 2003;34:267-73. PMID:
10.1038/ng1180. PMID:
12808457.
27.
Patti ME, Butte AJ, Crunkhorn S, Cusi K, Berria R, Kashyap S, Miyazaki Y, Kohane I, Costello M, Saccone R, Landaker EJ, Goldfine AB, Mun E, DeFronzo R, Finlayson J, Kahn CR, Mandarino LJ. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1.
Proc Natl Acad Sci U S A. 2003;100:8466-71.

Patti ME., Butte AJ., Crunkhorn S., Cusi K., Berria R., Kashyap S., Miyazaki Y., Kohane I., Costello M., Saccone R., Landaker EJ., Goldfine AB., Mun E., DeFronzo R., Finlayson J., Kahn CR., Mandarino LJ.. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1.
Proc Natl Acad Sci U S A 2003;100:8466-71. PMID:
10.1073/pnas.1032913100. PMID:
12832613.
28.
Phielix E, Schrauwen-Hinderling VB, Mensink M, Lenaers E, Meex R, Hoeks J, Kooi ME, Moonen-Kornips E, Sels JP, Hesselink MK, Schrauwen P. Lower intrinsic ADP-stimulated mitochondrial respiration underlies in vivo mitochondrial dysfunction in muscle of male type 2 diabetic patients.
Diabetes. 2008;57:2943-9.

Phielix E., Schrauwen-Hinderling VB., Mensink M., Lenaers E., Meex R., Hoeks J., Kooi ME., Moonen-Kornips E., Sels JP., Hesselink MK., Schrauwen P.. Lower intrinsic ADP-stimulated mitochondrial respiration underlies in vivo mitochondrial dysfunction in muscle of male type 2 diabetic patients.
Diabetes 2008;57:2943-9. PMID:
10.2337/db08-0391. PMID:
18678616.
29.
Quinn CE, Hamilton PK, Lockhart CJ, McVeigh GE. Thiazolidinediones: effects on insulin resistance and the cardiovascular system.
Br J Pharmacol. 2008;153:636-45.

Quinn CE., Hamilton PK., Lockhart CJ., McVeigh GE.. Thiazolidinediones: effects on insulin resistance and the cardiovascular system.
Br J Pharmacol 2008;153:636-45. PMID:
10.1038/sj.bjp.0707452. PMID:
17906687.
30.
Regensteiner JG, Bauer TA, Reusch JE. Rosiglitazone improves exercise capacity in individuals with type 2 diabetes. Diabetes Care. 2005;28:2877-83.

Regensteiner JG., Bauer TA., Reusch JE.. Rosiglitazone improves exercise capacity in individuals with type 2 diabetes.
Diabetes Care 2005;28:2877-83. PMID:
10.2337/diacare.28.12.2877. PMID:
16306548.
31.
Richter EA, Derave W, Wojtaszewski JF. Glucose, exercise and insulin: emerging concepts.
J Physiol. 2001;535:313-22.

Richter EA., Derave W., Wojtaszewski JF.. Glucose, exercise and insulin: emerging concepts.
J Physiol 2001;535:313-22. PMID:
10.1111/j.1469-7793.2001.t01-2-00313.x. PMID:
11533125.
32.
Rosenstock J, Niggli M, Maldonado-Lutomirsky M. Long-term 2-year safety and efficacy of vildagliptin compared with rosiglitazone in drug-naïve patients with type 2 diabetes mellitus. Diabetes Obes Metab. 2009;11:571-8.

Rosenstock J., Niggli M., Maldonado-Lutomirsky M.. Long-term 2-year safety and efficacy of vildagliptin compared with rosiglitazone in drug-naïve patients with type 2 diabetes mellitus.
Diabetes Obes Metab 2009;11:571-8. PMID:
10.1111/j.1463-1326.2008.01021.x. PMID:
19383032.
33.
Saltiel AR, Olefsky JM. Thiazolidinediones in the treatment of insulin resistance and type II diabetes.
Diabetes. 1996;45: 661-9.

Saltiel AR., Olefsky JM.. Thiazolidinediones in the treatment of insulin resistance and type II diabetes.
Diabetes 1996;45:661-9. PMID:
10.2337/diab.45.12.1661.
34.
Tonelli J, Li W, Kishore P, Pajvani UB, Kwon E, Weaver C, Scherer PE, Hawkins M. Mechanisms of early insulin-sensitizing effects of thiazolidinediones in type 2 diabetes.
Diabetes. 2004;53:1621-9.

Tonelli J., Li W., Kishore P., Pajvani UB., Kwon E., Weaver C., Scherer PE., Hawkins M.. Mechanisms of early insulin-sensitizing effects of thiazolidinediones in type 2 diabetes.
Diabetes 2004;53:1621-9. PMID:
10.2337/diabetes.53.6.1621. PMID:
15161771.
35.
Wolf G. Adiponectin: a regulator of energy homeostasis.
Nutr Rev. 2003;61:290-2.


Wolf G.. Adiponectin: a regulator of energy homeostasis.
Nutr Rev 2003;61:290-2. PMID:
10.1301/nr.2003.aug.290-292. PMID:
13677592.
36.
Zierath JR, He L, Gumà A, Odegoard Wahlström E, Klip A, Wallberg-Henriksson H. Insulin action on glucose transport and plasma membrane GLUT4 content in skeletal muscle from patients with NIDDM.
Diabetologia. 1996;39:1180-9.


Zierath JR., He L., Gumà A., Odegoard Wahlström E., Klip A., Wallberg-Henriksson H.. Insulin action on glucose transport and plasma membrane GLUT4 content in skeletal muscle from patients with NIDDM.
Diabetologia 1996;39:1180-9. PMID:
10.1007/BF02658504. PMID:
8897005.