INTRODUCTION
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has resulted in a global health emergency, and its heterogeneity driven by mutations and recombinations has instigated constant and long-term health threats [1,2]. SARS-CoV- 2 belongs to the Coronaviridae family and is a single-stranded RNA virus. Its viral spike glycoprotein (S protein) specifically binds to the human angiotensin-converting enzyme 2 (ACE2) receptor on the surface of alveolar epithelial cells for entry into host cells [3]. Essentially, the host serine protease TMPRSS2 cleaves the S protein and mediates membrane fusion for virus entry into host cells [4,5]. Infection with SARS-CoV-2 triggers a cytokine storm characterized by an uncontrolled systemic hyper-inflammatory response and primarily affects the respiratory system with diverse clinical symptoms such as high fever, cough, and severe pulmonary conditions [6,7]. Diarrhea, nausea, and vomiting have also been observed in a substantial proportion of patients, indicating that the gastrointestinal (GI) tract is associated with COVID-19 [8]. As ACE2 receptors are expressed not only in the upper respiratory tract, but also in the GI tract, the virus has been detected in lung, blood, and fecal samples from COVID-19 patients [9,10]. In fact, the virus has been detected in fecal samples at a higher level than in nasopharyngeal specimens of young COVID-19 patients [11]. Therefore, SARS-CoV-2 may affect different people in different ways throughout various organ systems by directly and indirectly deregulating the host immune systems [7].
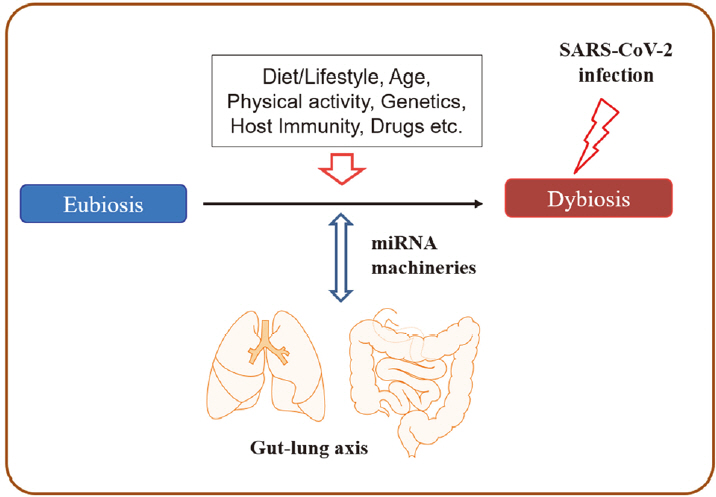
The human microbiome differs remarkably across age, sex, race, and ethnicity, suggesting that the specific and unique profiles of microbial population [12,13] perform many essential roles for human health [12]. In a healthy state, the human microbiota plays various roles, including energy recovery from primary and secondary metabolism, protection from pathogenic invasion, and modulation of inflammatory responses and the host immune system via interaction with host immune cells [14,15]. The importance of maintaining eubiotic conditions in microbial ecosystems is strongly associated with health and disease states, and it is known that multiple factors, such as lifestyle, diet, stress, and physical activity, can influence the status of the gut microbiome [14-17]. Growing evidence suggests that SARS-CoV-2 has been detected not only in the respiratory system (nasopharyngeal and mid-nasal specimens) but also in the GI tract (stool and rectal swabs) of COVID-19 patients. Infection of gut epithelial cells by SARS-CoV-2 can disrupt the gut microbiome, inducing dysbiosis, intestinal inflammation, and gastrointestinal symptoms [18,19]. Moreover, recent studies have shown that COVID-19 is often associated with altered gut microbiota composition, reflecting disease severity [20-23]. As growing evidence indicates the interaction between COVID-19 and host-microbiota homeostasis, researchers have been investigating the potential of manipulating gut microbiota with probiotics to enhance the host immune system [24,25]. The current review summarizes important findings on the direct association between SARS-CoV-2 infection and the gut-lung axis in addition to the pathological effects of dysbiosis on COVID-19 severity. We have also highlighted the potential roles of microRNAs (miRNAs) modulated by altered microbiota in COVID-19 pathogenesis. In conclusion, we suggest the implications of probiotics or miRNA-based therapies for the intervention and treatment of COVID-19.
METHODS
We searched for existing scientific literature using the keywords “COVID-19 or microbiota,” “microbiota or microRNA,” “COVID-19 or probiotics” in PubMed until March 31, 2021. Next, we reviewed the articles related to the alteration of human microbiota by SARS-CoV infection in respiratory and GI tract to understand the involvement gut-lung axis in COVID-19 pathogenesis. In addition, we have summarized the interaction between human microbiota and miRNA expression in the progression of COVID-19.
The altered gut and lung microbiota in the pathogenesis and prognosis of COVID-19
Multiple studies have shown the involvement of human microbiota in innate and adaptive immunity and respiratory infections [26]. The healthy human lung has low density but harbors a high microbial diversity with the most prevalent bacterial compositions of Bacteroides, Firmicutes, and Proteobacteria [27]. However, in case of COVID-19, the lung microbiota in bronchoalveolar lavage fluid of COVID-19 patients shows significant differences in microbiota composition, with enrichment of pathogenic and commensal bacteria, indicating microbial dysbiosis in COVID-19 [28]. In addition, Fat et al. have evaluated the lung microbiota in lung biopsies from 20 patients with fatal COVID-19 and observed that Acinetobacter, Brevundimonas, Burkholderia, and Chryseobacterium were commonly present with mixed fungal infections in these patients [29]. To date, few studies have analyzed the nasopharyngeal microbiota of patients with COVID-19 patients [30-32]. Hoque et al. have investigated the profiles and genomic determinants of the microbiome in nasopharyngeal specimens with and without COVID-19. They found that the typical composition of bacterial phyla is represented by Proteobacteria, Tenericutes, Actinobacteria, and Cyanobacteria in the COVID-19 metagenome [30] (Table 1). Although the dominant phyla showed no statistically significant differences among groups in concordance with another study [31], COVID-19 still affected the prevalence of certain microbes such as Proteobacteria and Cyanobacteria, suggesting that microbiomes in the respiratory tract might have a significant impact on the pathogenesis and severity of COVID-19 [30]. Moreover, Budding et al. have uncovered evidence for an association between decreased pharyngeal microbial diversity and SARS-CoV-2 infection and suggested an age-dependency of pharyngeal microbiota dysbiosis [32].
Along with lung microbiota, the composition and diversity of gut microbiota are also altered in COVID-19 patients, suggesting a bidirectional association between the respiratory and GI tract microbiota [18,33]. The commensal microbiota ecosystem in the gut is complex and dynamic, and plays an important role in regulating the host immune system and metabolism [34]. During COVID-19 infection, the disrupted gut microbiome contributes to the severity of COVID-19 by promoting gut permeability and systemic inflammation, which leads to impaired ACE2 expression [19,35]. Studies have shown that the gut microbiota of COVID-19 patients is characterized by decreased levels of beneficial bacteria with a predominance of opportunistic bacteria [20,21] (Table 1). A cross-sectional study evaluating the intestinal microbiota in fecal samples from COVID-19 patients and healthy controls has reported that the gut microbiota of COVID-19 patients is significantly dominated by opportunistic pathogens, such as Streptococcus, Rothia, Veillonella, and Actinomyces, with reduced bacterial diversity and beneficial symbionts compared with healthy controls [20]. Similarly, the gut microbiota of COVID-19 patients showed significant alterations during hospitalization, and intestinal dysbiosis was positively correlated with COVID-19 severity [21]. The baseline fecal abundance of opportunistic pathogens, such as Coprobacillus, Clostridium ramosum, and Clostridium hathewayi showed a positive correlation with COVID-19 severity. However, the anti-inflammatory bacterium, Faecalibacterium prausnitzii, was inversely correlated with the severity of COVID-19 in this study. In addition, Yeoh et al. have demonstrated that the dysbiotic gut microbiota composition is concordant with COVID-19 severity, which is associated with plasma concentrations of several cytokines, chemokines, and inflammation markers [22]. Even after recovery from COVID-19 and resolution of respiratory symptoms, gut microbiota dysbiosis was still detectable and persistent, suggesting implications for future immune-related health problems beyond COVID-19 [21,22]. Moreover, Gou et al. have suggested that the disrupted composition of gut microbiota may underlie the susceptibility of normal individuals to severe COVID-19, associated with an abnormal inflammatory status [36]. Prominently, these studies demonstrate a direct interaction between SARS-CoV-2 infection and microbiota dysbiosis of the respiratory and GI tracts. Therefore, severe COVID-19 patients may have disrupted the gut epithelial barrier, which may allow the virus to reach not only the gut-lung axis but also internal organs by entering the bloodstream [23].
The interaction of human microbiota and miRNAs in COVID-19 pathogenesis
The potential mechanism by which gut microbiota affects host pathophysiology is by precisely modulating the gene expression through miRNAs at the post-transcriptional level [37,38]. Nakata et al. have demonstrated that increased miR- 21-5p expression by commensal bacteria affects the intestinal epithelial permeability by regulating ADP ribosylation factor 4 [39]. Intestinal probiotics, Lactobacillus fermentum, and Lactobacillus salivarius can increase miR-155 and miR- 233 expression and enhance intestinal barrier function [40]. The exact mechanisms by which gut microbiota regulate miRNA expression remain largely unknown; however, different metabolites produced by gut microbiota may regulate the miRNA profiles of host cells. Notably, microbiota-derived extracellular vesicles harbor biologically active components, such as mRNAs and miRNAs, which may affect host gene expression [41]. Host miRNAs, such as fecal miRNAs mainly produced by intestinal epithelial cells, can be delivered to the gut microbiota and regulate the transcription and expression of microbial genes, shaping gut microbiota composition [42]. Collectively, these results indicate the implication of host miRNA expression for gut microbiota profiles and their bidirectional interaction in host homeostasis.
Previous studies have demonstrated that several viruses encode miRNAs that regulate host gene expression involved in promoting apoptosis in order to enhance their replication [43]. Aydemir et al. have identified 20 pre-miRNA candidates and 40 mature miRNAs encoded by SARS-CoV-2, which can regulate host gene expression by targeting NFKB, JAK/STAT, and TGFB signaling pathways [44] (Table 2). Therefore, viral miRNAs, such as SARS-CoV-mir-D8- 5p and SARS-CoV-mir-R1-5p, target many human genes involved in the transcription, metabolism, and immune systems, implicating the roles of miRNAs during viral infection [44,45]. Likewise, predicted 26 mature miRNAs from the SARS-CoV-2 genome might target human genes involved in innate antiviral immunity [46]. In SARS-CoV-2, small viral RNAs contribute to lung pathology by inducing pro-inflammatory cytokines, whereas their antagomirs specifically reduced the inflammatory lung pathology, highlighting the potential role of the small viral RNA antagomiRs in eliciting direct antiviral effects [47].
Host miRNAs can interfere with viral replication both directly and indirectly by inducing antiviral reactions in the progression of viral infection [48] (Table 2). It has been demonstrated that the abundance and profile of miRNAs are associated with the severity and mortality of COVID-19 in aged patients, indicating the essential roles of host cellular miRNAs in the pathogenesis of COVID-19 [49]. Likewise, the low expression and lack of differential expression of miRNAs have been predicted to promote susceptibility of lung epithelial cells to SARS-CoV-2 infection [50]. Arisan et al. have identified 7 key miRNAs (miR-8066, miR-5197, miR- 3611, miR-3934-3p, miR-1307-3p, miR-3691-3p, and miR- 1468-5p) with significant links to viral pathogenicity and host responses [51]. When evaluating the miRNA expression pattern in peripheral blood, miR-16-2-3p, miR-6501-5p, and miR-618 were the most upregulated miRNAs in COVID-19 patients compared to the control group. In contrast, the expression of other miRNAs, such as miR-183-5p, miR-627- 5p, and miR-144-3p, was significantly reduced in patients compared to healthy donors, and were associated with the dysregulation of immune function through differential miRNA expression profile [52]. Recently, the potential functions of 13 host miRNAs in SARS-CoV-2 infection have been systematically reviewed, suggesting their potential roles in the interaction between miRNAs and viral activity [53]. Moreover, several studies have investigated host miRNAs that directly target the expression of ACE2 and TMPRSS2, which are critical for viral entry and insertion. For example, miR-1246, miR-200c-3p, and miR-125a-5p were predicted to regulate ACE2 expression levels and their associated pathways [54-56], while let-7a-5p and let-7d-5p were reported to negatively correlate with TMPRSS2 expression [56-58]. These results demonstrate the impact of host cellular miRNAs on SARS-CoV-2 infection and COVID-19 pathogenesis.
DISCUSSION
In this review, we summarize the current literature to date that provides evidence for the interaction of lung and gut microbiota in the pathogenesis and prognosis of COVID-19, with implications for modulating miRNA expression by altered microbiome signatures (Figure 1).
The diversity and abundance of microbiota signatures among individuals strongly affect human health and disease- related inflammatory and metabolic conditions [12-16]. In a healthy state, the human microbiota plays various roles, including energy recovery from metabolism, protection from pathogenic invasion, and modulation of the host immune system [14,15]. Several factors can modify human microbiota, including host genetics, lifestyle, diet, age, stress, physical activity, and antibiotics, and they continuously affect the healthy microbiota throughout the life of the host [14-17]. In particular, the frequency and duration of physical activity have strong evidence for a positive association with gut microbiome diversity [15]. In addition, host cells affect the composition and productivity of gut microbiota by releasing nonspecific factors (e.g., antimicrobial peptides, secreted immunoglobulin A and mucins, and epithelial barrier) and specific factors, such as miRNAs [17,59]. For example, hsamiR- 515-5p and hsa-miR-1226-5p can enter bacterial cells and have a specific effect on gut bacterial growth [17]. In addition, the gut microbiota interacts with the host through various ligands (e.g., pathogen-associated molecular patterns) and produces bioactive metabolites, including shortchain fatty acids, anti-inflammatory and anti-proliferative lipids, essential vitamins (vitamins B and K), hormones, and serotonin [14]. Consequently, gut microbiota can influence host pathophysiology, and host factors reciprocally shape the bacterial ecosystem across individuals.
Growing evidence on host-microbe interactions indicates that microbiota is an essential mediator in communication between the gut and other organs and maintaining human health via the gut microbiota-miRNA interactions [60,61]. Interplays between microbiota and miRNAs have been reported in several lung diseases [62], implicating the beneficial effects of probiotics on the modulation of miRNAs. Thus, the administration of probiotics may function through several signaling pathways, resulting in the prevention and treatment of various pathological conditions [63]. As microbiome dysbiosis is linked with the severity of COVID-19, the implication of probiotics in modulating the severity has been suggested as a promising weapon against COVID-19 [24,25,64]. Several studies focusing on the gut microbiome and probiotics in COVID-19 have been conducted, and several registered clinical trials have focused on the usage of probiotics in COVID-19 [24].
MicroRNAs produced by all living organisms and viruses are well-conserved and regulate the expression of their target genes [65]. Although the exact mechanisms of host and viral miRNAs in SARS-CoV-2 infection are not well-defined, it is evident that virus-encoded miRNAs and host cellular miRNAs are associated with the initiation and severity of COVID-19. SARS-CoV-2 encoded miRNAs alter host gene expression to create a favorable environment for viral infection. Interestingly, host miRNAs can play dual roles as both antiviral and proviral factors [45]. The number of host miRNAs targeting the SARS-CoV-2 genome sequence inhibits viral replication by regulating innate antiviral immunity [53]. Other host microRNAs can increase the stability of the viral genome and inhibit decay, which is beneficial for viral replication and propagation [66]. All these results indicate the potential impact of miRNAs on SARS-CoV-2 and host interplay, suggesting the development of therapeutic approaches for miRNAs against COVID-19. Therefore, miRNA-based antiviral therapies, such as miRNA mimics or inhibitors, can be used for the intervention and treatment of COVID-19 [67,68].
The United States Centers for Disease Control and Prevention (CDC) has reported that 45% of COVID-19 patients requiring hospitalization are ≥ 65 years old, and the highest mortality and morbidity against COVID-19 have been reported in older patients with underlying chronic diseases associated with inflammation (https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/index.html). Elderly patients with certain medical conditions may be highly vulnerable to the infection with SARS-CoV-2 because they are associated with altered gut microbiota and the integrity of epithelial barriers [69]. In addition, the expression of ACE2, which is critical for viral interaction with host cells, has been shown to increase with age, accounting for increased susceptibility to older patients [70]. Moreover, the abundance and profile of miRNAs have also been associated with the severity and mortality of COVID-19 in aged patients, indicating the essential roles of host cellular miRNAs in the pathogenesis of COVID-19 [49].
In conclusion, the human microbiota-miRNA axis can be used as a promising therapeutic approach for the management of COVID-19. Therefore, further studies are essential to investigate the exact molecular mechanisms of how altered human microbiota regulate the miRNA expression profile and how these deregulated miRNA profiles mediate viral infection through host-microbe interactions.