Effect of high-intensity exercise and high-fat diet on lipid metabolism in the liver of rats
Article information
Abstract
[Purpose]
This study investigated the effects of high-intensity exercise (Ex) and high dietary fat intake on lipid metabolism in the liver of rats.
[Methods]
Male Sprague-Dawley rats were randomly assigned to one of the four groups (n=10 per group) that were maintained on a normal diet (ND) or high-fat diet (HFD) consisting of 30% fat (w/w), with or without exercise on a treadmill at 30 m/min and 8% grade) for 4 weeks (i.e., ND, ND+Ex, HFD, and HFD+Ex groups).
[Results]
Body weight (p<.001), total plasma cholesterol (TC) (p<.001), triglyceride (TG) (p<.05), and liver TG levels (p<.05) were increased in the HFD group relative to the ND groups, and serum glucose (p<.05), insulin (p<.05), homeostatic model assessment of insulin resistance (HOMA-IR) (p<.01), and liver TG levels (p<.01) were also higher in the HFD group compared to the ND+Ex group. Plasma free fatty acid was elevated in the HFD+Ex group compared to the HFD group (p<.01). With the exception of acetyl coenzyme A carboxylase, the expression of lipid metabolism-related genes in the liver was altered in the Ex groups compared to the control group (p<.05), with genes involved in lipolysis specifically up regulated in the HFD+Ex group compared to the other groups.
[Conclusion]
Vigorous exercise may increase glucose utilization and fat oxidation by activating genes in the liver that are associated with lipid metabolism compared to that in animals consuming a HFD without exercise. Therefore, high intensity exercise can be considered to counter the adverse effects of high dietary fat intake.
INTRODUCTION
Excess fat in the diet leads to metabolic disorders including obesity, hypertension, hyperinsulinaemia, and diabetes that can be debilitating for individuals and also constitute a public health challenge [1,2]. These diseases are characterized by chronically increased circulation of free fatty acids (FFAs) and elevated insulin secretion, which are linked to pathogenic mechanisms [3] such as altered oxidation of various biomolecules that can impair cellular functions and lead to apoptosis [4,5].
Lipid synthesis is regulated by insulin and nutrient availability, with higher insulin levels stimulating lipogenesis and gluconeogenesis in the liver [6] and muscle tissue [7]. Insulin induces the upregulation of lipid metabolism-related genes, leading to increased synthesis of saturated and monounsaturated fatty acids and triglycerides (TGs). High-fat diet (HFD) increases hepatic lipid content [8,9], with higher fat weight and greater incidence of fatty liver observed at only 11 days after commencing an HFD, with alterations in liver lipid metabolism reported after 1-2 weeks [9,10]. Excess free fatty acid accumulation from a HFD also enhances glucose utilization via the glycolytic pathway, and insulin resistance in peripheral cells causing hyperinsulinaemia, which could therefore explain the development of fatty liver [11].
Regular, moderate exercise can reduce blood glucose and improve plasma lipoprotein profiles after 2 weeks in humans and in animal models [12,13]. However, even a single bout of high-intensity exercise (Ex) can have negative consequences on liver functions, including metabolic pathways and detoxification [8,14], while some reports have suggested that strenuous exercise can prevent fat accumulation in the liver and muscle tissue in rats [15]. It was hypothesized that the combination of an HFD and Ex can influence glucose and FFA levels via secretion of insulin and regulation of lipid metabolism. The present study investigated the effects of Ex and an HFD on lipid profiles and the expression of lipid metabolism-related genes in the liver of rats.
METHODS
Animals, diet, and exercise
The animal protocol for this study was approved by the Animal Care and Use Committee of Dankook University. Male Sprague-Dawley rats (n=40; Charles River, St.-Constant, QC, Canada), 4 weeks of age and weighing 180-200 g, were housed individually in 20.7×35×17 cm cages in a controlled environment at 22±1°C on a 12:12 h light/dark cycle (7:00-19:00 h) with access to food and tap water ad libitum. After a 1-week acclimatization, rats were randomly divided into four groups (n=10 per group): normal diet (ND) or HFD without exercise, and a ND or HFD with treadmill exercise (ND+Ex and HFD+Ex, respectively). The HFD consisted of 30% fat (w/w), compared to 3.5% fat from total energy in the ND, based on modified recommendations of the American Institute of Nutrition [16]. After 8 weeks on the diets, animals in the Ex group were subjected to the exercise protocol, while sedentary rats were allowed to continue feeding for 4 weeks without exercise. Rats were familiarized with the exercise regimen by running on a motorized treadmill for 25-30 min/day, 5 days/week at a speed of 20 m/min and an incline of 8% during the first week. The regimen was then switched to 60 min/day, 5 days/week for 4 weeks at a speed of 30 m/min and an incline of 8%. The exercise intensity was based on previous studies [17,18], which estimated for maximal oxygen consumption of approximately 70% for the rats.
Sample collection and measurement of blood lipid profiles
Rats were sacrificed with an anesthetic after fasting for 18 h. A blood sample was drawn from the artery and the liver was immediately dissected, rinsed with saline, flash-frozen in liquid nitrogen, and stored at -80°C until analysis.
Plasma total cholesterol (TC), triglyceride (TG), and serum high-density lipoprotein cholesterol (HDL-C) levels were measured using a commercial enzyme-linked immunosorbent assay kit (ELI Tech, Seoul, Korea). Serum low-density lipoprotein cholesterol (LDL-C) was calculated using the Friedewald formula [19]. Fasting plasma glucose concentrations were determined by the glucose oxidase method using YSI 2300 STAT Plus glucose analyzer (YSI Life Sciences, Springfield, VA, USA). Serum insulin levels were measured by radioimmunoassay using a commercial kit (Millipore, St. Charles, Missouri, USA) with rat insulin as a standard, which was extracted from pancreatic tissue samples by overnight sonication at 4°C. Homeostatic model assessment of insulin resistance (HOMA-IR) was calculated and FFA level was measured as previously described [20]. Liver TG content was estimated from the amount of glycerol released after ethanolic KOH hydrolysis using a spectrophotometer at 470nm.
RNA isolation and reverse transcription (RT)-PCR
Total RNA was extracted from 40-50 mg of liver tissue using Trizol reagent (Invitrogen, Carlsbad, CA, USA) and purified using a Micro-to-Midi Total RNA Purification System (Bioneer, Daejeon, Korea). The purity of the RNA was confirmed by gel electrophoresis and the concentration was determined using a spectrophotometer at an absorbance of 260 nm. The expression of sterol regulatory element binding protein (SREBP)-1C, acetyl coenzyme A carboxylase (ACC), fatty acid synthase (FAS), carbohydrate-responsive element-binding protein (ChREBP), stearoyl coenzyme A desaturase (SCD)-1, and carnitine palmitoyltransferase (CPT)-1α mRNA was assessed by semi-quantitative RT-PCR on a thermal cycler (Thermo Hybaid, Middlesex, UK), using the forward and reverse primer sets as shown in Table 1, which was used to normalize the expression levels of the other genes. After a hot start at 94°C for 2 min, the cycling conditions were as follows: 94°C for 45 s, 55-65°C for 45 s, and 72°C for 30 s, followed by 72°C for 5 min. The reaction products were resolved by gel electrophoresis and visualized using a UV illuminator/imaging system (UNOCK-800, Seoul Korea); quantification of signal intensity was performed by densitometry using Image J software (National Institutes of Health, Bethesda, MD, USA).
Western blotting
Liver tissue samples were homogenized in ice-cold homogenization buffer and resolved by polyacrylamide gel electrophoresis, transferred to polyvinylidene fluoride membranes, and incubated at room temperature in phosphate-buffered saline for 12 h with an antibody against ACC1 (1:500; BD Transduction Laboratories, Lexington, KY, USA) followed by an anti-rabbit secondary antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA). The signal was detected by enhanced chemiluminescence (Amersham Pharmacia Biotech, Piscataway, NJ, USA) and densitometry was performed using Image J software program (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
Data were expressed as mean±SEM. Data were analyzed by one-way analysis of variance with the Tukey post-hoc test using the SPSS statistical program (version 13.0 SPSS for Windows; Inc., Chicago, IL, USA). Differences were considered statistically significant at p<0.05.
RESULTS
TC, TG, and plasma glucose and insulin levels are elevated by high fat intake, while FFA level is increased by exercise
Sedentary animals on a HFD had a body weight that was approximately 10.4% higher than groups on a ND (p<.001) and these rats also had higher liver weight than those fed a ND. Plasma TC (p<.001) and TG (p<.05), and liver TG levels (p<.05) were higher in the HFD group than in the two ND groups, while plasma HDL-C (p<.001) level was elevated in animals fed a HFD and/or groups that exercised compared to those consuming a ND with no exercise. High dietary fat intake alone, and not vigorous exercise alone, altered plasma glucose (p<.05), insulin (p<.05), and HOMA-IR (p<.01), which were higher in the HFD group than in the ND+Ex group. In contrast, FFA level was higher in rats that exercised (ND+Ex group, p<.05 and HFD+Ex group, p<.01) than in those that did not exercise; the ND and HFD groups (Table 2).
High dietary fat intake leads to upregulation of mRNA expression of lipogenesis-related genes, while mRNA expression of lipolysis-related genes is stimulated by exercise
The mRNA expression level of SREBP-1C was highest in rats with high dietary fat intake that also exercised; the HFD+Ex group (p<.001). FAS mRNA expression was up-regulated in rats that exercised and/or were fed a ND, and it was lower in rats consuming a HFD without exercise than in those on a ND with exercise (p<.001). The mRNA transcript level of ChREBP was higher in rats that exercised than in sedentary rats (p<.05); among the sedentary rats, the expression was higher in the HFD group than in the ND group. Exercise also decreased SCD-1 mRNA expression, which was lower in the ND+Ex and HFD+Ex groups than in the ND group (p<.05). The combination of HFD and exercise- as compared to either factor by itself-increased CPT-1α mRNA expression, which was higher in the HFD+Ex group than in the HFD (p<.01) or ND+Ex group (p<.05) (Fig. 1).

Expression of lipid metabolism-related genes in the liver. Data are presented as means±standard deviation (n=10 per group): normal diet (ND); high-fat diet (HFD); exercise (Ex); (A): sterol regulatory element binding protein (SREBP)-1C; (B) acetyl coenzyme A carboxylase (ACC); (C) fatty acid synthase (FAS); (D) carbohydrate regulatory element-binding protein (ChREBP); (E) stearoyl coenzyme A desaturase (SCD)-1; and (F) carnitine palmitoyltransferase (CPT)-1α relative to the expression level of β-actin. The mean of triplicate reactions is shown. *p<0.05, **p<0.01, ***p<0.001
There was no difference in the expression of ACC across groups at the mRNA level, but ACC protein synthesis in the liver was higher in rats on a HFD with or without exercise than in those fed a ND (p<.05), and it was highest in the HFD+Ex group compared to the HFD group (p<.05) (Fig. 2).
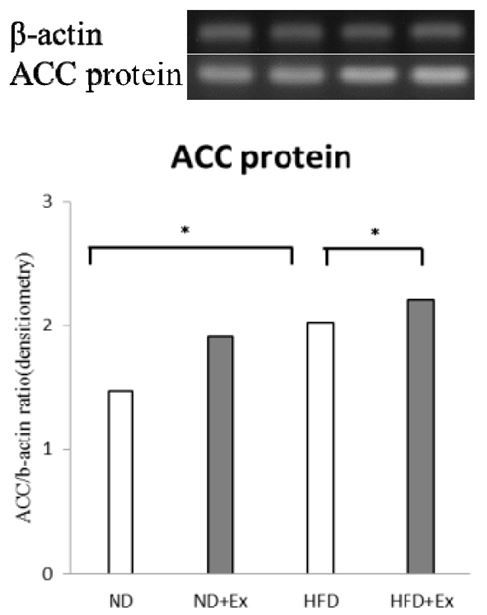
Protein expression of acetyl coenzyme A carboxylase (ACC) in the liver. Data are presented as means±standard deviation SEM (n=10 per group). The experimental groups were ND, normal diet; ND+Ex, normal diet with high-intensity exercise; HFD, high-fat diet; HFD+Ex, high-fat diet with high-intensity exercise: Acetyl Co-A carboxylase (ACC), *p<0.05
DISCUSSION
Various metabolic disorders are induced by a HFD, and here it was observed that high dietary fat intake resulted in body mass gain and hepatic fat accumulation in rats, with increased plasma TC and TG levels in the HFD groups relative to animals on a ND with or without exercise. Liver TG, as well as glucose, insulin, and HOMA-IR were also higher in the HFD group than in the ND+Ex group, indicating that diet plays a greater role than exercise in these physiological variables. These results are consistent with other studies that found high plasma TC and TG [21], and glucose [22] levels due to lipid overload in the liver as a result of high dietary fat intake.
Although it is widely accepted that regular exercise has beneficial effects in an organism, including reduced blood glucose utilization and increased fat utilization, it is less clear whether the same is true for exercise that is performed at high intensity over a prolonged period [23]. Moderate exercise stimulates lipolysis, and enhances circulating FFA level and insulin activity [4,17]. In animal studies, vigorous exercise has shown adverse effects, and the cellular mechanisms that determine whether the resultant lipid accumulation is well- tolerated [18] or toxic [24] are poorly understood. Animals in the exercise groups (ND+Ex and HFD+Ex) were similar to the control (ND) group in terms of body weight, plasma TC and TG, and liver TG; however, HDL-C levels were higher. Plasma FFA concentration is regulated by a balance between lipolysis, glucose uptake by the blood, and β-oxidation in the liver. In this study, the FFA level was higher in the HFD+Ex group than in the HFD group, and also in the ND+Ex group relative to the HFD group, suggesting that lipolysis was stimulated by exercise, thereby increasing the FFA level in the plasma. This dysregulation of fatty acid homeostasis may promote insulin resistance and other complications, such as aberrant fatty acid uptake or oxidation [25]. The decreased liver TG level in the HFD+Ex group indicates that exercise, even at high intensity and in conjunction with high fat consumption, is conducive to maintaining body weight and normal lipid profiles [26].
SREBP-1C is a transcription factor that mediates the biosynthesis of cholesterol and fatty acids by regulating the expression of genes involved in lipogenesis, such as ACC and FAS [27,28], and by co-ordinately stimulating TG synthesis in other tissues [12,29]. Chronic activation of lipogenesis- related genes induced by a HFD may promote liver TG accumulation and decrease the availability of FFA [30]. The transcription factor, ChREBP also modulates the expression of lipogenic genes in the liver for the synthesis of TGs from carbohydrates [31], and regulates the transcription of genes encoding enzymes required for the entry of glucose into the glycolytic pathway [32]. ChREBP overexpression in liver tissue results in hyperglycemia [33].
SREBP-1C expression was highest in rats that consumed a diet high in fat and engaged in intense exercise, while FAS mRNA and ACC protein were up regulated in animals that exercised regardless of the diet compared to sedentary controls. The transcript level of ChREBP was also higher in the exercise groups relative to animals consuming the same diet but without exercise. In a previous study, ChREBP expression had beneficial effects on insulin signaling and blood glucose level, possibly resulting from an exercise-induced increase in glycolysis [34]. The upregulation of ChREBP observed here suggests that exercise stimulates glycolysis and not lipogenesis, while hepatic fat accumulation was inhibited by the increase in SREBP-1C and FAS mRNA and ACC protein expression in both exercise groups [34].
SCD-1 is the rate-limiting enzyme in the biosynthesis of saturated fat-derived monounsaturated fats [35], and its expression is decreased upon diet-induced weight gain; moreover, the down-regulation of hepatic SCD-1 expression as a result of exercise could reflect reduced lipogenic activity [36], and may be associated with lower liver TG and increased plasma FFA levels [35,37]. Consistent with the earlier findings, SCD-1 transcript expression was higher in the sedentary rats compared to those that performed strenuous exercise.
CPT-1α catalyses the transfer of fatty acids from CoA to carnitine, an essential step in the β-oxidation of fatty acids and their conversion into energy in the liver. In animals with high dietary fat intake combined with an intense exercise regimen, CPT-1α expression was higher than in animals subjected to either one of these conditions. CPT-1α expression was reported to increase after exercise, and was associated with increased fatty acid oxidation [6,38]. The upregulation of CPT-1α in the HFD+Ex group suggests that exercise counters the effects of high fat consumption and prevents hepatic fat accumulation by stimulating β-oxidation of fatty acids.
CONCLUSION
A diet with high fat content had adverse effects in rats, resulting in weight gain and dysregulation of glucose and lipid metabolism in the liver. Vigorous exercise reversed these effects, improving lipid profiles despite high fat consumption. Although elucidation of the precise mechanisms requires a more extensive study, these findings can help in developing strategies aimed at managing metabolic disorders resulting from high dietary intake of fat.

