INTRODUCTION
For thousands of years, ginseng (Panax Ginseng Meyer) has been known to be a medicinal plant, showing a variety of effects on the human body and is a substance that helps maintain homeostasis in the living body through an "adaptogen effect" [12]. In recent years, it has become of the most commonly used functional herbs in dietary supplements [23]. The reported medicinal effects have been being supported by clinical studies in the fields of physiology, biology, biochemistry, pharmacology, and pathology [2].
Saponin, the main active ingredient, is responsible for a variety of proven effects of ginseng, and is usually referred to as ginsenosides. This name refers to a glycoside isolated from ginseng, which distinguishes it from saponin and from other plants. There are close to 60 species of ginsenosides [20], and of those ginsenosides, Rg1 belongs to the PT (protopanaxtriol) class and is one of the typical ginsenosides abundant in Panax Ginseng [8].
According to previous or ongoing studies of the pharmacological activities of ginsenosides, Rg1 treatment reportedly strengthens the hippocampus which plays an important role in memory and learning [16, 26]. It has also been reported to have various effects including enhancement of immune function, inhibition of platelet aggregation, stimulation of protein synthesis (mouse brain), anti-fatigue, anti-stress, anti-inflammation, promotion of hepatocellular proliferation and DNA synthesis, promotion of nerve cell survival, stimulation of cholesterol (activating LDL receptor), and stimulation of adrenocorticotropin secretion [2].
However, in the field of exercise science, there are few studies that have evaluated the effect of Rg1 intake. Furthermore, previous studies reported that it is difficult to draw any clear conclusion on the relationship between ginseng and improvement of exercise capacity [7]. The main limitations for the clinical study are harvest time, and varietal species caused by varying cultivation, which leads to frequently changing profiles of ginsenosides [8].
A number of limitations were likely present because no specific dose and concentration of single or combined ginsenosides has been clearly validated. In order to solve these problems, it is necessary to examine the effect of each specific ginsenoside in ginseng which acts on human body in relation to exercise capacity and associated metabolic processes [3,4,5]. Through this, medicinal or functional foods which can enhance physical health and function can be developed, and specific methods for an ergogenic aid can be presented.
No positive effect of long-term treatment with Rg1 in human muscle strength and anaerobic power has been observed, though there were positive effects on antioxidant enzymes, immune responses, and VO2max [8,15,24,25]. In addition, the activity of citrate synthase (CS), which is a typical aerobic enzyme used as an indicator of mitochondrial function, increased [8]. It is thought to be very difficult to make a clear conclusion of improved aerobic capacity based on the changes of only one indicator enzyme. Moreover, adverse effects of a long-term treatment with ginseng, such as high blood pressure, digestive disorders, insomnia, and nervousness have been reported [13]. Thus, considering its use for long-term condition management or as a temporary coping mechanisms in athletes, short-term intake (1-3 times/24 hours) should be assessed. Compared with other tissues, skeletal muscle is the most able to adapt to changes or stimulation. It reacts in a variety of different morphological and biochemical ways by exposure to a certain environment, regular aerobic and anaerobic exercises, and nutritional or pathological stimuli. Because such changes rely on the mobilization pattern of the muscle fibers, it is necessary to selectively observe a muscle group. In this study, aerobic, plantaris muscle fibers from a dominant muscle group were chosen to observe bigger morphological and biochemical changes after 6 weeks of swimming.
Endurance training was a 6-week course of swimming using Sprague-Dawley rats, followed by acute uptakes of Rg1 three times within 24 hours. Aerobic capacity was tested by swimming time to exhaustion. Immediately after the measurement, the plantaris was extracted and mass changes, CS encoding mRNA production, and CS activity were analyzed. Through these results, the additive effects of a short-term intake of Rg1 on enhanced aerobic exercise capacity acquired through a long-term swimming training were examined.
METHODS
Experimental animal
Experimental animals were 8-week-old male rats of the Sprague-Dawley lineage. Seven rats were assigned to each group and they went through adaptation periods for cage and swimming exercise for 10 days before the onset of treatment. After that, except for the subjects eliminated during the 6-week treatment period, the final 26 rats were divided into four groups, placebo (NP, n=6), Rg1 (NRG, n=6), exercise+placebo (EP, n=7), and exercise+Rg1 (ERG, n=7). The whole study was conducted after the animal care and the treatment procedures were approved by Kyung Hee University Animal Ethics Committee (KHUASP-15-21).
Swimming exercise
Each swimming exercise was 50 minutes, five days a week, for 6 weeks, following a method used in a previous study with some modifications [18]. The water bath was 43cm in depth and was 102cm in diameter and 47cm in height, and the water temperature was between 28-32‚ÑÉ.
Acute uptake of Rg1
Rg1 was provided Kyung Hee Hanbang Bio, Inc., (Korea) and used in a powder form. The powder was dissolved in water and administered orally using a sonde. The dose was 50mg/500g body weight [22]. It was administered three times within 24 hours including once in the morning and once in the afternoon of the next day when the exercise groups (EP, ERG) completed each of the exercises, and 30 minutes prior to measuring aerobic exercise capacity the following morning.
Swimming time to exhaustion test for the assessment of aerobic capacity
In order to evaluate aerobic exercise capacity, swimming time to exhaustion was measured by a method used in a previous study after modifications [9]. The testing environment was the same as the environment used for the swimming exercise and a weight, corresponding to 3% of the body weight, was attached to the tail. Exhaustion was determined as the time when the animal was unable to rise to the surface within 10 seconds after sinking to the bottom.
Muscle sampling and RT-PCR
Immediately after completion of the aerobic capacity test, the animal was decapitated with a guillotine and the plantaris was sampled. Its weight was measured immediately after isolation and was quick-frozen in liquid nitrogen and stored at -70℃ until analysis. 300 μl PureHelixTM RNA Extraction Solution (NanoHelix, Korea) was added to a microtube to which approximately 80~100 mg muscle tissues were added and completely grinded. Then an additional 700 μl PureHelixTM RNA Extraction Solution was added to the sample following 15~30 seconds of vortexing and stored at room temperature for 5 minutes. In order to remove non-homogenized materials, it was centrifuged at 12,000 rpm for 5 to 10 minutes at 4℃. 200 μl chloroform was added to every 1ml RNA extraction solution, followed by inversion for 15 to 30 seconds and the sample was kept for 2-15 minutes at room temperature and then centrifuged again at 12,000 rpm for 10 minutes at 4℃. 500 μl of the upper layer of the supernatant containing RNA was taken from the microtube which was separated by centrifugation and transferred to a new sterilized microtube. Isopropyl alcohol, corresponding to 0.6% of the volume transferred to the microtube, was added, and then it was kept on ice for 5-10 minutes, followed by centrifugation at 12,000 rpm for 10-15 minutes at 4℃. After centrifugation, the supernatant was discarded and the pellet was rinsed by vortexing with 75% ethanol made of RNA free-water, followed by centrifugation at 12,000 rpm for 5 minutes at 4℃. For complete removal of ethanol, the sample was air-dried and then an appropriate amount of 0.1% diethyl pyrocarbonate (DEPC) water was added. Finally, it was kept at 55~60℃ for 10-15 minutes to extract the RNA. The prepared samples were used to measure purity and concentration with a spectrophotometer using the absorbance at A260 and A280. For measuring the expression level of CS mRNA, Helix CriptTM One-step Reverse Transcription-Polymerase Chain Reaction (RT-PCR) Kit (NanoHelix, Korea) was used for cDNA synthesis of each gene and RT-PCR was performed by mixing a fixed amount of primers, specimen samples and DEPC water, and placed the reaction mixtures in a PC3-20 primer Thermal Cycler (Astec, USA). β-actin, a housekeeping gene, was used as a reference gene for quantifying the amount between samples and the primer sequences used in the RT-PCR analysis are as shown in <Table 1>.
Determination of citrate synthase activity
Analysis of CS activity in the plantaris was performed following the methods used in a previous study [21]. Enzyme activity was calculated by the dilution factor and the molar extinction coefficient (13.6). The activity unit was indicated as μmol/min/g.
Statistical analysis
A statistical software package SPSS PC+ for Windows (version 22.0) was used in this study, and two-way ANOVA was applied to each dependent variable to test for differences depending on Rg1 intake and exercise status. If the primary effects among groups were statistically significant, the difference within each group was verified by conducting a t-test for independent samples. The level of statistical significance (α) for all statistical processes was set to less than 0.05.
RESULTS
Body mass and muscle weight changes
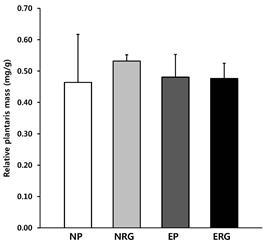
During the 6-week experimental period, the body weight of the experimental animals was measured once a week at the same time of the day and the results are shown in <Fig. 1>. There was no significant difference in body weight between groups. The mass of the plantaris was expressed as relative weight by incorporating body weight (mg/g). The effect of 6-week swimming exercise and short-term Rg1 treatment on muscle mass was compared and are shown in <Fig. 2>, the mean was 0.489mg/g and neither the effect of interaction nor the primary effect between groups was observed based on treatment.
Fig.¬Ý1.
Body weight changes. It shows the patterns of body weight changes of rats in each treatment group for six weeks and there is no significant difference between groups.NP: placebo, NRG: Rg1, EP: exercise+placebo, ERG: exercise+Rg1. Mean±SD. P<.05.

Fig.¬Ý2.
Muscle weight change in plantaris. This graph shows the changes in mass of the plantaris after 6 weeks between groups, indicated as the relative muscle mass considering the body weight (mg/g), in which no group-specific changes in the mass of the plantaris were observed. NP: placebo, NRG: Rg1, EP: exercise+placebo, ERG: exercise+Rg1. Mean±SD. P<.05.
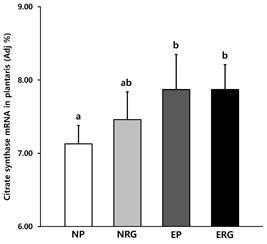
Expression of citrate synthase mRNA
Changes in the expression level of CS mRNA are shown in <Fig. 3>. For the level of CS mRNA expression in plantaris, the primary effect appeared in the exercise groups EP and ERG (P<.001). The primary effect was analyzed and the results showed that EP and ERG showed a significantly higher level of mRNA expression than the placebo group NP (P<.01, P<.001).
Fig.¬Ý3.
Expression of citrate synthase mRNA in plantaris. It shows the level of CS mRNA expression appeared in the plantaris after 6-weeks of swimming and acute administration of Rg1, in which exercise and Rg1 treatment on its own or in combination resulted in significant changes in the expression level compared with NP group. However, there was no additive effect by combined treatment. Same acronyms indicate no significant difference between groups by t-test. NP: placebo, NRG: Rg1, EP: exercise+placebo, ERG: exercise+Rg1. Mean±SD. P<.05.
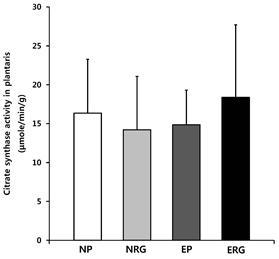
Citrate synthase activity
For the level of CS activity, neither the effect of the interaction, nor the primary effect between groups, was observed depending on treatment <Fig. 4>.
Fig.¬Ý4.
Citrate synthase activity in plantaris. Citrate synthase activity appeared in the plantaris after 6-week swimming and acute administration of Rg1, in which no significant changes were observed in any treatment group. NP: placebo, NRG: Rg1, EP: exercise+placebo, ERG: exercise+Rg1. Mean±SD. P<.05.
Swimming time to exhaustion
The results of the swimming time to exhaustion test are shown in <Fig. 5>, and the primary effect depending on treatment appeared in EP and ERG (P<.001). EP showed 1,655±36sec and ERG showed 2,213±70sec, which exhibited 2.35 and 3.14 times longer exercise time, respectively, compared with NP (P<.01, P<.001).
Fig.¬Ý5.
Swimming time to exhaustion. Aerobic exercise capacity obtained through swim to exhaustion time test conducted after 6-week swimming and acute administration of Rg1, in which acute administration of Rg1 alone didn’t change, whereas the exercise group and combined treatment group exhibited improved exercise capacity. The additive effect of Rg1 was verified in the combined treatment group, in particular. Same acronyms indicate no significant difference between groups by t-test. NP: placebo, NRG: Rg1, EP: exercise+placebo, ERG: exercise+Rg1. Mean±SD. P<.05.

DISCUSSION
In this study, the effect of acute uptake of Rg1 following 6-weeks of swimming exercise on the capacity of aerobic exercise was examined in 8-week-old male SD rats.
Body weight was measured once a week in the same time of the day during the 6-week experimental period except for the adaptation period, in which no significant difference between groups was found during the treatment period depending on treatment. Therefore, it is thought that 6-week swimming exercise and short-term intake of Rg1 did not affect the total body mass of SD rat at all.
Due to the muscle fiber type composition of the plantaris that it is mainly of type II muscle fibers, it was expected that the changes in the capacity of aerobic exercise caused by long-term swimming were likely to be greater than other hind-limb muscles, which was the reason why the plantaris was chosen for this study [19]. There was no significant difference in the mass of the plantaris relative to the total body weight based on treatment. The probability that the mass of the plantaris changed significantly by aerobic exercise, especially by the modified 6-week swimming exercise protocol employed in this study [18] has never been suggested in the previous studies, which is consistent with our findings. No previous studies have reported changes in the mass of the plantaris caused by intake of ginsenoside including Rg1 and in particular, it is thought that morphological changes including additional mass were unlikely to be induced by acute uptake of Rg1 following swimming. In conclusion, long-term aerobic exercise, immediately followed by acute administration of Rg1 did not result in morphological changes in the plantaris. And this result is grounds for an interpretation that the increase in the exercise duration time, which is to be discussed later, was caused by changes in biochemical factors.
The level of CS mRNA expression was measure by RT-PCR and the results showed that the level of mRNA expression was significantly higher in EP and ERG than in NP. The increase of CS mRNA expression in the plantaris caused by long-term aerobic exercise has been confirmed by a previous study [19]. In addition, a tendency toward an increase of mRNA expression caused by Rg1-intake alone was observed, but without significance. So far, no previous study examined the level of CS mRNA expression depending on Rg1 treatment. However, the results presented in this study are consistent with previous studies that long-term exercise stimulated and increased CS mRNA expression in the plantaris, which was detected even in six weeks after the exercise, and Rg1-intake alone was likely to affect CS mRNA expression. However, considering that there was a tendency of reduced or constant level of CS mRNA expression after a certain amount of time as shown in previous studies [19], the point that the combined treatment did not bring additive effects can be interpreted as that it suggests the parts to be studied in the future.
There was no significant difference in the CS activity in the plantaris muscle tissue depending on treatment. CS activity has been used as a biomarker representing the mitochondrial capacity and naturally representative of the capacity for endurance exercise of the skeletal muscle caused by long-term endurance exercise in both animals and humans. In a previous study targeting human, Rg1 (capsule 5mg) isolated from Panax Notoginseng was treated two times, one hour before cycling and the previous night, and then the CS activity was shown to be elevated in the blood in three hours after cycling [8]. However, in this study, which measured the CS activity in the plantaris muscle tissue, ERG showed the highest activity, but it was lower than the activity shown in previous studies [12,19], and there was no significant difference between treatments. This suggests that though the dose of Rg1 treated within 24 hours stimulated Rg1 production, the protein synthesis caused by mRNA production may have been affected by a time factor, thus it did not influence the enzyme activity enough.
In swimming time to exhaustion, results showed that compared with NP and NRG, EP and ERG exhibited significance differences with swimming for longer periods of time (more than 2-folds and 3-folds longer, respectively). ERG showed the additive effect by acute intake of Rg1 in addition to the changes caused by long-term exercise. A previous study conducted on cycling time to exhaustion in humans using Rg1 treatment and the results of the study showed that the placebo group showed 31.8±5.0min and Rg1 treatment group showed 38.3±6.7min, which was significant, and the short-term treatment of Rg1 was found to give a positive effect on aerobic capacity [8]. In another previous study on ginseng treatment, graded exercise was performed after 3-weeks of treatment with Panax Ginseng in humans, and the results showed that although no significant difference was observed, the Panax Ginseng treatment group showed longer exercise time [1]. Apart from this, many previous studies related to ginseng and exercise reported that administration of ginseng or Rg1 could give a positive impact on improvement of exercise capacity [3-7,17]. The results from many studies were obtained using long-term intake of ginseng or ginsenoside, whereas short-term administration studies like this one were extremely rare.
To sum up, acute intake of Rg1, showed an additive effect on improvement of aerobic exercise capacity, which suggests that many attempts for developing new methods for utilizing efficacy ingredients mainly from ginseng and ginsenoside are necessary in the future. Considering that there are few studies available, and that the reported results are not consistent, not only are the mechanisms of action of a single active ingredient such as Rg1, but also the field research such as doses and timing are required additionally.
CONCLUSION
In this study, whether acute uptakes of Rg1 following 6-week swimming can give any additive benefit on aerobic exercise capacity in SD rats was examined. Although CS mRNA production increased in exercise group (EP) and combined treatment group (ERG), there was no significant differences in the CS activity, which indicated that production of a specific mRNA and the protein it encodes do not have a direct relationship to the temporal and positional differences. However, acute uptakes of Rg1 following long-term swimming was shown to provide an additive effect on aerobic exercise capacity when using the swimming time to exhaustion test. This posits that a short-term intake of Rg1 can be used as ergogenic aids. It also notes the necessity of additional research on the mechanism of action and on the details of application protocols.








