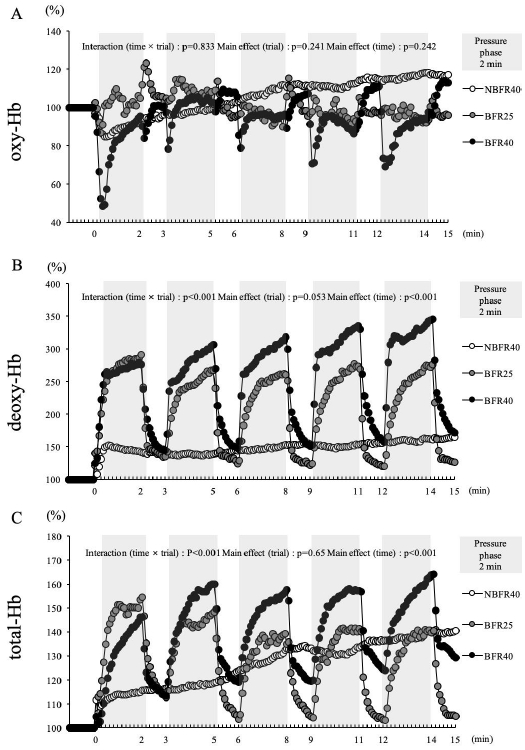
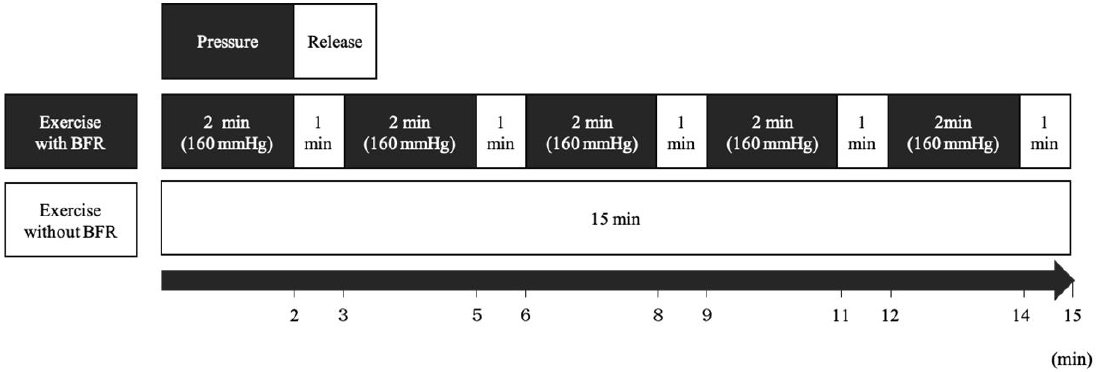
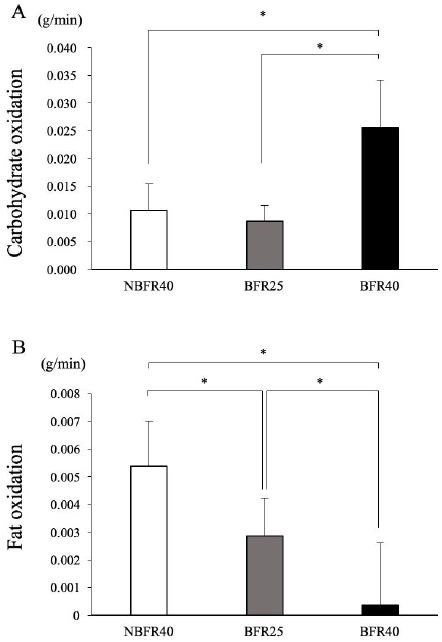
1.
Kraemer WJ, Ratamess NA, French DN. Resistance training for health and performance.
Curr Sports Med Rep. 2002;1:165-71.
. PMID:
10.1249/00149619-200206000-00007.
2.
Yasuda T, Brechue WF, Fujita T, Sato Y, Abe T. Muscle activation during low-intensity muscle contractions with varying levels of external limb compression. J sports sci med. 2008;7:467.
.
3.
Takarada Y, Sato Y, Ishii N. Effects of resistance exercise combined with vascular occlusion on muscle function in athletes.
Eur J Appl Physiol. 2002;86:308-14.
. PMID:
10.1007/s00421-001-0561-5.
4.
Abe T, Fujita S, Nakajima T, Sakamaki M, Ozaki H, Ogasawara R. Effects of low-intensity cycle training with restricted leg blood flow on thigh muscle volume and VO2 max in young men. J sports sci med. 2010;9:452.
.
5.
Sudo M, Ando S, Kano Y. Repeated blood flow restriction induces muscle fiber hypertrophy.
Muscle Nerve. 2017;55:274-6.
. PMID:
10.1002/mus.25415.
6.
Ladlow P, Coppack RJ, Dharm-Datta S, Conway D, Sellon E, Patterson SD. Low-load resistance training with blood flow restriction improves clinical outcomes in musculoskeletal rehabilitation: A single-blind randomized controlled trial.
Front Physiol. 2018;9:1269.
. PMID:
10.3389/fphys.2018.01269.
7.
Yasuda T. Fukumura K, Tomaru T, Nakajima T. Thigh muscle size and vascular function after blood flow-restricted elastic band training in older women.
Oncotarget. 2016;7:33595-607.
. PMID:
10.18632/oncotarget.9564.
8.
Yasuda T, Loenneke JP, Thiebaud RS, Abe T. Effects of blood flow restricted low-intensity concentric or eccentric training on muscle size and strength.
Plos one. 2012;7:e52843.
. PMID:
10.1371/journal.pone.0052843.
9.
Hamlin MJ, Marshall HC, Hellemans J, Ainslie PN, Anglem N. Effect of intermittent hypoxic training on 20km time trial and 30s anaerobic performance.
Scand J Med Sci Sports. 2010;20:651-61.
. PMID:
10.1111/j.1600-0838.2009.00946.x.
10.
Sundberg C, Eiken O, Nygren A, Kaijser L. Effects of ischaemic training on local aerobic muscle performance in man.
Acta Physiol Scand. 1993;148:13-9.
. PMID:
10.1111/j.1748-1716.1993.tb09526.x.
11.
Ohno H, Shirato K, Sakurai T, Ogasawara J, Sumitani Y, Sato S. Effect of exercise on HIF-1 and VEGF signaling.
J Phys Fit Sports Med. 2012;1:5-16.
. PMID:
10.7600/jpfsm.1.5.
12.
Lundby C, Jacobs RA. Adaptations of skeletal muscle mitochondria to exercise training.
Experimental physiology. 2016;101:17-22.
. PMID:
10.1113/EP085319.
13.
Conceicao MS, Gaspari AF, Ramkrapes APB, Junior EMM, Bertuzzi R, Cavaglieri CR. Anaerobic metabolism induces greater total energy expenditure during exercise with blood flow restriction.
PLoS One. 2018;13:e0194776.
. PMID:
10.1371/journal.pone.0194776.
14.
Corvino RB, Rossiter HB, Loch T, Martins JC, Caputo F. Physiological responses to interval endurance exercise at different levels of blood flow restriction.
Eur J Appl Physiol. 2017;117:39-52.
. PMID:
10.1007/s00421-016-3497-5.
15.
de Oliveira MF, Caputo F, Corvino RB, Denadai BS. Short-term low-intensity blood flow restricted interval training improves both aerobic fitness and muscle strength.
Scand J Med Sci Sports. 2016;26:1017-25.
. PMID:
10.1111/sms.12540.
16.
Kelly LP, Basset FA. Acute Normobaric Hypoxia Increases Post-exercise Lipid Oxidation in Healthy Males.
Front Physiol. 2017;8:293.
. PMID:
10.3389/fphys.2017.00293.
17.
Joyner MJ, Casey DP. Regulation of increased blood flow (hyperemia) to muscles during exercise: a hierarchy of competing physiological needs.
Physiol Rev. 2015;95:549-601.
. PMID:
10.1152/physrev.00035.2013.
18.
Takano H, Morita T, Iida H, Asada K, Kato M, Uno K. Hemodynamic and hormonal responses to a short-term low-intensity resistance exercise with the reduction of muscle blood flow.
Eur J Appl Physiol. 2005;95:65-73.
. PMID:
10.1007/s00421-005-1389-1.
19.
Hamaoka T, McCully KK, Quaresima V, Yamamoto K, Chance B. Near-infrared spectroscopy/imaging for monitoring muscle oxygenation and oxidative metabolism in healthy and diseased humans.
J Biomed Opt. 2007;12:062105.
. PMID:
10.1117/1.2805437.
20.
Horiuchi M, Okita K. Blood flow restricted exercise and vascular function.
Int J Vasc Med. 2012;2012:543218.
. PMID:
10.1155/2012/543218.
21.
Tran TK, Sailasuta N, Kreutzer U, Hurd R, Chung Y, Mole P, Kuno S, Jue T. Comparative analysis of NMR and NIRS measurements of intracellular PO2 in human skeletal muscle.
Am J Physiol. 1999 Jun;276:R1682-90.
. PMID:
10.1152/ajpregu.1999.276.6.R1682.
22.
Nioka S, Kime R, Sunar U, Im J, Izzetoglu M, Zhang J. A novel method to measure regional muscle blood flow continuously using NIRS kinetics information.
Dyn Med. 2006;5:5.
. PMID:
10.1186/1476-5918-5-5.
23.
Ganesan G, Cotter JA, Reuland W, Cerussi AE, Tromberg BJ, Galassetti P. Effect of blood flow restriction on tissue oxygenation during knee extension.
Med Sci Sports Exerc. 2015;47:185-93.
. PMID:
10.1249/MSS.0000000000000393.
24.
Suga T, Okita K, Morita N, Yokota T, Hirabayashi K, Horiuchi M. Intramuscular metabolism during low-intensity resistance exercise with blood flow restriction.
J Appl Physiol. 2009;106:1119-24.
. PMID:
10.1152/japplphysiol.90368.2008.
25.
Spranger MD, Krishnan AC, Levy PD, O'Leary DS, Smith SA. Blood flow restriction training and the exercise pressor reflex: a call for concern.
Am J Physiol Heart Circ Physiol. 2015;309:H1440-52.
. PMID:
10.1152/ajpheart.00208.2015.
26.
Mendonca GV, Vaz JR, Teixeira MS, Gracio T, Pezarat-Correia P. Metabolic cost of locomotion during treadmill walking with blood flow restriction.
Clin Physiol Funct Imaging. 2014;34:308-16.
. PMID:
10.1111/cpf.12098.
27.
Neto GR, Santos HH, Sousa JB, Junior AT, Araujo JP, Aniceto RR. Effects of high-intensity blood flow restriction exercise on muscle fatigue.
J Hum Kinet. 2014;41:163-72.
. PMID:
10.2478/hukin-2014-0044.
28.
Suga T, Okita K, Takada S, Omokawa M, Kadoguchi T, Yokota T. Effect of multiple set on intramuscular metabolic stress during low-intensity resistance exercise with blood flow restriction.
Eur J Appl Physiol. 2012;112:3915-20.
. PMID:
10.1007/s00421-012-2377-x.
29.
Suga T, Okita K, Morita N, Yokota T, Hirabayashi K, Horiuchi M. Dose effect on intramuscular metabolic stress during low-intensity resistance exercise with blood flow restriction.
J Appl Physiol. 2010;108:1563-7.
. PMID:
10.1152/japplphysiol.00504.2009.
30.
Pearson SJ, Hussain SR. A review on the mechanisms of blood-flow restriction resistance training-induced muscle hypertrophy.
Sports Med. 2015;45:187-200.
. PMID:
10.1007/s40279-014-0264-9.
31.
Cumming KT, Paulsen G, Wernbom M, Ugelstad I, Raastad T. Acute response and subcellular movement of HSP27, alphaB-crystallin and HSP70 in human skeletal muscle after blood-flow-restricted low-load resistance exercise.
Acta Physiol (Oxf). 2014;211:634-46.
. PMID:
10.1111/apha.12305.
32.
Smiles WJ, Concei├¦├Żo MS, Telles GD, Chacon-Mikahil MP, Cavaglieri CR, Vechin FC, Libardi CA, Hawley JA, Camera DM. Acute low-intensity cycling with blood-flow restriction has no effect on metabolic signaling in human skeletal muscle compared to traditional exercise.
Eur J Appl Physiol. 2017;117:345-58.
. PMID:
10.1007/s00421-016-3530-8.
33.
Amann M, Eldridge MW, Lovering AT, Stickland MK, Pegelow DF, Dempsey JA. Arterial oxygenation influences central motor output and exercise performance via effects on peripheral locomotor muscle fatigue in humans.
J Physiol. 2006;15:937-52.
. PMID:
10.1113/jphysiol.2006.113936.
34.
Miura H, McCully K, Nioka S, Chance B. Relationship between muscle architectural features and oxygenation status determined by near infrared device.
Eur J Appl Physiol. 2004;91:273-8.
. PMID:
10.1007/s00421-003-0964-6.