The effect of regular Taekwondo exercise on Brain-derived neurotrophic factor and Stroop test in undergraduate student
Article information
Abstract
[Purpose]
The purpose of this study was to investigate the effect of Taekwondo exercise on Brain-derived neurotrophic factor and the Stroop test in undergraduate students.
[Methods]
Fourteen male subjects participated in this study. They were separated into a Control group (N = 7) and an Exercise group (N = 7). Subjects participated in Taekwondo exercise training for 8 weeks. They underwent to Taekwondo exercise training for 85 minutes per day, 5 times a week at RPE of 11~15. The taekwondo exercise training comprised an aerobic exercise (20min) mode and a dynamic exercise (65min) mode. All data were analyzed by repeated measures two-way ANOVA.
[Results]
There were no significant differences in the physical characteristics of the subjects. Although weight and BMI showed a tendency to decreased in the exercise group (EG). Also, neurotrophic factors (BDNF, NGF, IGF-1) were not significantly different after 8 weeks in the two groups. However, BDNF and IGF-1 showed a tendency to increase in the exercise group (EG). Finally, the Stroop test (word, color) results were significantly different(p < .05) in the exercise group (EG).
[Conclusion]
These finding suggest that 8 weeks of regular Taekwondo exercise training may increase cognitive functions (Stroop test). However the training did not statistically affect neurotrophic factors (BDNF, NGF, IGF-1) in undergraduate students.
INTRODUCTION
Taekwondo is a traditional Korean martial art. Lately, a variety of research has been carried out in the field of taekwondo studies. The martial art was designated a national Korean sport in 1971, and in 1972, the Kukkiwon, the headquarters of taekwondo, was founded to promote the sport internationally. After a long campaign, taekwondo was finally designated an official Olympic discipline in the Sydney Olympics, in 2000. At present, taekwondo, as the representative sport of Korea, is trained by millions of practitioners in 188 countries around the world. However, taekwondo is not simply a sport and educational tool in middle and high schools, it continues to expand in the sphere of the humanities and physical an education. Lately, taekwondo has contributed to other specialized educational activities, such as creative performance, after school education, and school sports clubs [12]. As such, taekwondo enriches our lives as a sport for all in the 21th century. Moreover, it has positive effects on practitioners’ health and helps to promote the national image of Korea. In fact, taekwondo itself has been established as the representative national brand of Korea. It has been proven that regular exercise promotes health, physical condition, and muscle strength in addition to preventing obesity, hypertension, and diabetes. The focus of previous studies was mostly on conduct and therapy. However, a change in direction occurred recently toward studying the effects on brain activity. Exercise creates neuronal networks, which increases the thinking process of the brain and enables better memory. Exercising increases blood flow in the brain and, therefore, causes the growth of neuronal networks and the improvement of cognitive functions [10]. Recently, to scientifically demonstrate the effect on the brain functions, computer tomography, emission tomography (PET), and functional magnetic resonance imaging (fMRI) were applied. In addition, such imaging techniques were reportedly employed in the disciplines of psychology and medicine, sports psychology, and physical education.
Active research across several fields using cognitive questionnaires and biochemical tests has recently investigated neuroplasticity-related substances (brain-derived neurotrophic factor [BDNF], nerve growth factor [NGF], and tropomyosin receptor kinase B, etc.) to prove the effect of exercise on the improvement of brain function. Of these, BDNF reportedly has a positive effect on neurogenesis, neurodegeneration, and neural plasticity and also promotes memory and learning [23]. BDNF-a protein associated with neuronal activity in the spinal cord and peripheral nervous system-is activated in the hippocampus, cortex, cerebellum, and basal forebrain, which are portions of the brain that play a central role in learning, memory, and higher-order thinking. It also plays a particularly important role in long-term memory [4]. Moreover, insulin-like growth factor-1 (IGF-1)-one of the factors that control the expression of the BDNF gene-increases the expression of neurotrophic factors as a result of aerobic exercise, stimulates neurogenesis, and helps the brain recover from injury. Furthermore, vascular endothelial growth factor (VEGF) and IGF-1 both play an important role in cytogenesis and angiogenesis, which is the process of capillary production from already existing blood vessels [24]. Current exercise-related research mostly focuses on the topic of regular exercise and consists not only of studies on disease prevention and treatment but also of studies on brain function. However, with regard to current taekwondo-related research, despite worldwide awareness of its excellence and importance, most research in the field of natural sciences has focused on stamina as well as obesity-related and hyperlipidemic factors in taekwondo novices. The scope of study topics has only begun to expand recently. Accordingly, there are very few studies on changes in brain activation, cognitive function, and neurotrophic factors as a result of taekwondo. Therefore, changes in brain neurotrophic factor activity and cognitive function with regular Taekwondo exercise training remain unknown. The aim of this study was to examine the effect of Taekwondo exercise on Brain-derived neurotrophic factor and the Stroop test in undergraduate students.
METHODS
Subjects
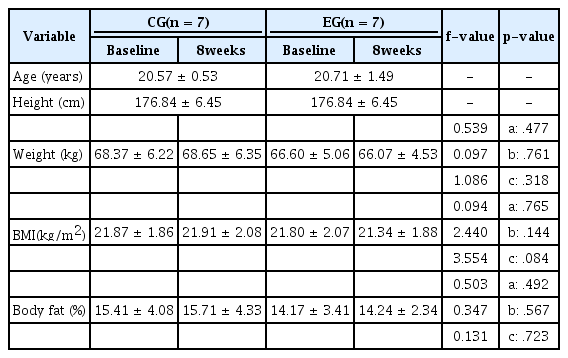
A total of 14 undergraduate male subjects at Y University participated in the experiment. The subjects were divided into two groups: control group (CG, n = 7) and exercise group (EG, n = 7). The two groups were randomly selected. All participants had no cardiac medical conditions and also had no other diseases. They voluntarily agreed to participate in the experiment after being briefed on the purpose. The physical characteristics of the subjects are described in Table 1.
Anthropometric assessment and body fat measurement
Height and weight were measured as the physical characteristics, and body fat percentage was measured using an impedance analyzer (GAIA 359 Plus, Jawon medical, Korea). Measurements were performed before and after the 8-week Taekwondo exercise training program. Body mass index (BMI) was defined as the weight in kilograms divided by the square of the height in meters.
8-week Taekwondo exercise training
The taekwondo exercises program consisted of largely aerobic training (20 minutes) and dynamic exercise training (60 minutes). The aerobic exercise training consisted of a 400m run on the track within 20 minutes (RPE 11-13), followed by jogging. The dynamic exercise training consisted of largely isotonic Mitt-(target)-exercises and isometric exercises, followed by run jumping exercises, divided into 3 exercise mode movements.
The following 4 kicking techniques were executed on the Mitt-target: front-kick, roundhouse-kick, turn-kick, and spinning-back-kick. 10 sets with 10 repetitions were executed during a time span of 20 minutes (RPE 13-15). Before the Mitt-target exercises, 2 sets, with 10 repetitions, of front-kicks, roundhouse-kicks, and side-kicks were executed in the air, without target contact. After each set, the kicking leg was rested and stretched for 10-15 seconds. Finally, 6m jumping exercises separated into 2 sets, with 10 repetitions, over 10 minutes (RPE 13-15), were performed. After the aerobic and dynamic exercise, stretching was performed for 5 minutes. A 5 minute rest was allowed between each step. The total daily exercise time consisted of 85 minutes and was performed over 5 days.
Measurement of neurotrophic factor
Blood samples were drawn from the forearm vein after a 12-hour fast. Samples were centrifuged at 3,000 rpm for 10 minutes, stored at -80℃, and directly analyzed.
Serum BDNF levels were analyzed with an R & D system (Minneapolis, Minn., USA) kit and serum IGF-1 levels were analyzed with a Quantikine sandwich enzyme immunoassay (DG 100, R&D Systems, USA). The results were obtained by measuring the optical density at 450 nm. Also, biochemical analysis of serum NGF level was performed using enzyme-linked immunosorbent assays for quantitative detection of human NGF (Abcam, MA, USA).
Stroop test
Cognitive tests were performed using the Stroop-test [22]. The Stroop test(word, color, and color-word) consists of three sessions, all of which are variations of the same theme; participants are required to read out loud, as quickly and accurately as possible, from a list of items for 45 s. The word tests, requires that subjects read from a page containing three printed words: red, green, or blue. The words are printed in black ink and are listed in random fashion in five columns of 20 words each. No word is allowed to follow itself in a column. The color test, similarly consists of 100 items written as XXXX and printed in either red, green, or blue ink. The subject is required to states the color of the ink for each of the series of four X`s. The color-word test, consists of the same 100 words (red, green, or blue) as the first test, but is printed in colored ink, The color of the ink and the printed word are never the same (for example, the word ‘‘blue’’ could be printed in red or green ink, but never in blue ink). The subject is required to say the color of the ink the word is printed in, not the printed word itself. For the three tests, a higher score reflects a better performance [7].
Statistical analysis
For all data, the mean and standard deviation were calculated using SPSS for Windows ver. 14.0 (SPSS, Chicago, IL, USA). Repeated measures two-way ANOVA was used to control for differences between groups, test periods, physical characteristics, and biochemical blood component changes caused by Taekwondo exercise training. All statistical significance level (α) were set at .05.
RESULTS
Physical characteristics
Subjects’ physical characteristics (weight and BMI, %fat) were recorded before and after the 8 weeks of regular Taekwondo exercise. The results are presented in Table 1.
There were no significant differences in weight, BMI, and %fat score among the groups or test periods, and there was no interaction effect between the group and the test period.
Change in neurotrophic factor
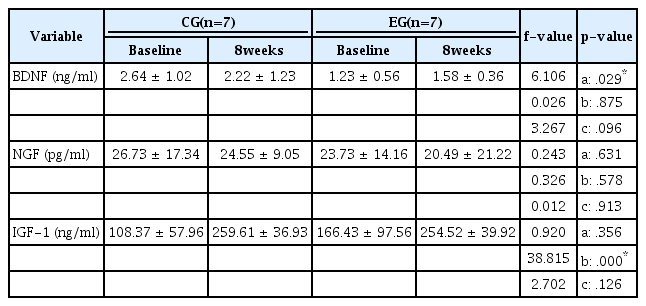
The 8 weeks of regular Taekwondo exercise resulted in little changes in the neurotrophic factors (serum BDNF, NGF, and IGF-1). The results are presented in Table 3. There were no significant differences or interaction effects of BDNF, NGF, and IGF-1 among the groups, between test periods, or between group and test period. However, BDNF and IGF-1 levels showed a tendency to increase in the exercise group (EG).
Change in Stroop- test(cognitive function)
The Stroop-test(cognitive function) score was recorded before and after the 8 weeks of regular Taekwondo exercise. The results are presented in Table 4.
There were significant differences in word test score among the groups (F = 12.395, p < .05) and test periods (F = 6.558, p < .05). There was an interaction effect between group and time (F = 13.682, p < .05), and further analysis of the interaction effect revealed a significant increase (p < .05) in the EG after the 8 weeks of regular Taekwondo exercise.
There were no significant differences in color test score among the groups, but there was a significant difference between the test periods (F = 30.259, p < .05). There was an interaction effect between group and time (F = 7.282, p < .05), and further analysis of the interaction effect revealed a significant increase (p < .05) in the EG after 8 weeks of regular Taekwondo exercise.
There were no significant differences in word-color test score among the groups, and there was no interaction effect between group and test period.
DISCUSSION
Brain derived neurotrophic factor (BDNF), is a member of the neurotrophic family, which promotes neuronal survival and synaptic plasticity [1], it is an essential neurotrophin that is also intimately connected with central and peripheral molecular processes of energy metabolism and homeostasis, and could play a crucial role in these the induction of these mechanisms [11].
The effect of regular exercise on BDNF levels is highly influenced by variables such as exercise type, intensity, and frequency. Exercise types can be broadly categorized into strength (resistance) training and aerobic training. In all previous studies on resistance training, regular training did not increase resting BDNF levels [8,15,18,25]. With regard to different periods of exercise for resistance training studies, Goekint et al. [8] and Levinger et al. [15] evaluated a 10-week resistance training duration, Schiffer et al. [18] used a 12-week period, and Yarrow et al. [25] used a 5-week duration.
The reasons that resistance training did not change resting BDNF levels in these studies may be related to the exercise type (resistance) and duration (5-12 weeks), but Knaepen et al. [11] suggested that the lack of change was because the frequency in the above studies (three times per week) was less than five times a week.
Aerobic training studies can be divided into those that found aerobic training to significantly increase resting BDNF levels [2,20,26] and those that found no significant differences [6,19]. Because the various studies used different subjects, training periods, frequencies, and exercise intensities, it is difficult to identify consistency in terms of the resting BDNF-increasing effects of regular training. However, high intensity (heart rate reserve [HRR], 65-85%) and frequency (4-7 times per week rather than 2-3 times per week) were common in those studies that showed an increase in resting BDNF levels resulting from regular aerobic training.
As mentioned above, the exercise type-dependent (aerobic or resistance) increase in BDNF levels showed a greater effect on aerobic training over resistance training, while aerobic training seemed to be influenced to a greater extent by exercise intensity and frequency rather than by duration.
The exercise intensity and frequency in our study were as high as the intensity (rating of perceived exertion, 13-15; HRR > 60%) and frequency (more than five times per week) used in previous studies that found an increase in resting BDNF with regular aerobic training [2,20,26]. However, the exercise group (EG) in our study did not show significantly increased resting BDNF levels over the 8-week training period. This is thought to be because the exercise type used in our study was complex (aerobic, isometric, and power), consisted of 75% dynamic exercise and 25% aerobic exercise, and comprised less aerobic exercise than the 100% aerobic exercise that was previously found to increase resting BDNF levels.
With the current lack of studies on the effect of complex exercise on BDNF, our study results are worth presenting. Although the differences were not statistically significant, we did find an increasing pattern. In the future, it will be necessary to perform additional research with more subjects to increase the statistical power. If it is proven that BDNF levels significantly increase due to taekwondo, a ripple effect can be expected on taekwondo-related research and the taekwondo industry.
NGF (nerve growth factor) plays an important role in the sustenance of sympathetic and sensory neurons as well as in biological activities, including cell growth [21].
It is difficult to find results from studies on NGF in healthy individuals. Schulz et al. [19], who assessed regular exercise-related results, found that when patients with multiple sclerosis (MS) performed 8 weeks of ergometer-measured aerobic exercise, the training group did not show significant differences in NGF levels. Additionally, Bansi et al. [3] did not find a significant change in resting NGF levels for middle-aged patients with MS who performed 3 weeks of regular exercise (aquatic vs. overland). However, a domestic study showed a significant taurine administration-dependent increase in NGF expression in the hippocampi of rats that underwent 8 weeks of swimming exercise [14].
In our study as well, neither group (control [CG] or EG) showed a significant increase in resting NGF levels after 8 weeks; instead, both groups showed a slight decrease-a finding that is consistent with those of previous studies [19]. Considering the findings of the studies mentioned above [3, 19], it is difficult to increase NGF levels with short-term training; as such, it will be necessary to investigate the changes in NGF levels with long-term training in future studies.
IGF-1 (insulin growth factor-1) is defined to a neurotrophic factors, BDNF gene expression and increases the generated neurons by aerobic exercise training. Also, it serves to help recover from brain damage [24].
The IGF-1 results also varied by exercise type and duration, with one-time exercise and 12 weeks of complex exercise causing a significant increase [13,17]. Meanwhile, 8 weeks of aerobic training and 10 weeks of strength training did not cause a statistically significant increase in IGF-1 levels [6,8]. In the past, IGF-1 was studied as a growth hormone; however, it has been recently studied in relation to neurotrophic factors such as BDNF [2,6,8,13,17]. Accordingly, in our study, we mostly referenced the results for IGF-1 that were related to BDNF. In our results, there was an increasing trend with 8 weeks of regular taekwondo exercise in the EG; however, neither group (CG or EG) showed a significant interaction, and hence, it is difficult to determine whether there was an effect following 8 weeks of exercise.
The benefits of physical activity are not limited to improved cardiovascular function. The benefits include better brain function, protection against neurodegeneration, and improved neuroplasticity, cognitive function, and mental health. These effects of exercise have been proven by various studies.
Exercise-related studies using the Stroop color and word test can be divided into those involving one-time exercise [7,17] and those involving regular training [5,9,16]. Regular aerobic and resistance training has been proven to improve cognitive function in various previous studies. The improvement in cognitive function from regular training is associated with BDNF levels. In a report by Griffin et al. [9], the importance of cognitive function and BDNF was emphasized since BDNF levels increased with cognitive function after 5 weeks of aerobic exercise. In the Stroop test in our study, which was conducted to identify changes in cognitive function following 8 weeks of training, we found a statistically significant interaction in the word and color test for the CG and EG groups. The EG group showed a significant increase in word and color scores after 8 weeks compared to the CG group, clearly demonstrating increased cognitive function. These results are consistent with those of previous research [13], and is believed to be due to the fact that the EG in our study showed a trend of increased resting BDNF levels after 8 weeks of taekwondo even though the difference did not reach statistical significance.
CONCLUSIONS
In conclusion, 8-weeks of regular Taekwondo exercise training (aerobic and dynamic exercise) did not significantly affect brain-derived neurotrophic factor, however the Stroop-test (cognitive function) showed a significant (p < .05) increase. These findings are therefore limited, and the effects of regular Taekwondo exercise on neurotrophic factors in undergraduate students were not conclusively proven. Future research will need to focus on related areas.